All steps of preparation of fabric for dyeing.
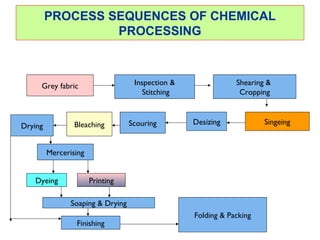
- 1. PROCESS SEQUENCES OF CHEMICAL PROCESSING Grey fabric Inspection & Stitching Shearing & Cropping SingeingDesizingScouringBleachingDrying Mercerising Dyeing Printing Soaping & Drying Finishing Folding & Packing
- 2. Preparation / Pretreatment Processes used to remove impurities from fibres to make it dye able or printable. Natural fibers and synthetic fibers contain primary impurities that are contained naturally, and secondary impurities that are added during spinning, knitting and weaving processes. Textile pretreatment is the series of cleaning operations .All impurities which causes adverse effect during dyeing and printing is removed in pretreatment process.
- 3. Objective of Pretreatment: To Convert fabric from hydrophobic to hydrophilic state. To remove dust, dirt etc from the fabric. To achieve the degree of desire whiteness.
- 4. 4 Preparation Following are the steps to be taken before going wet processing : Grey inspection Stitching Shearing and cropping Singeing
- 5. Desizing Scouring Bleaching Mercerization Typically a woven cotton fabric would be prepared by sequence of process as shown. In case of knitting sizing step is not involved Preparation/Pretreatment Singeing
- 6. SHEARING & CROPPING In shearing ,the fibres are cut in an angular manner on the surface of the fabric itself, resulting in a soft feel Singeing and shearing are almost the same in effect. In singeing the fibres in the interlacement of the fabric are burnt by flames whereas in shearing the fibres are cut in an angular manner on the surface of the fabric itself, resulting in a soft feel.
- 7. Singeing Burning of protruding fibres from the surface of fabric or yarn - A mechanical process singeing refers to the burning-off of Loose fibres not firmly bound into the yarn and/or fabric structure. Textiles materials are most commonly singed in woven or knitted fabric form or in yarn form. Makes fabrics smooth Prevents pilling Improves luster
- 8. Advantages of Singeing Singeing of a fabric is done in order to obtain a clean fabric surface which allows the structure of the fabric to be clearly seen. Fabrics, which have been singed, soil less easily than un-singed fabrics. The risk of pilling, especially with synthetics and their blends, is reduced in case of singed fabrics. randomly protruding fibres are removed in singeing which could cause diffused reflection of light.
- 9. Necessity of Singeing in Textile Cotton materials are valued for their smooth appearance. After the formation of fabric it has a fuzzy or hairy appearance due to projecting fibers, thus affecting the luster and smoothness Unsigned fabrics are soiled easily The protruding fibers obstruct the subsequent dyeing and printing process Goods which are to be mercerized are signed to maximize the luster In fabrics of polyester and cellulosic fiber blends singeing is the best method to control pilling, sometimes double singeing is done to minimize the pilling.
- 10. Singeing Process Singeing process is as follows: To produce a smooth surface finish on fabrics made from staple fibers first the fabric surfaces are brushed lightly to raise the unwanted fiber ends. Then the fabric is singed with or passed over heated copper plates or open gas flames. The fiber ends burn off. The fabric is moved very rapidly, and only the fiber ends are destroyed. As soon as the fabric leaves the singeing area, it enters a water bath or desizing bath. This stops any singeing afterglow or sparks that might damage the cloth.
- 14. Bio polishing It is an enzyme treatment designed to improve fabric quality and provide following advantages: Improve pilling resistance A clear, lint and fuzz-free surface structure Improved drape and softness The effect are durable
- 15. Process Cellulase enzyme is used Enzyme dosage 1-2% owf pH 4.5-5.5 Temperature 40-55o C Time 30-60 min. It is similar to singeing as it also provide similar effect on the fabric.
- 16. Fig. 7. Carbomatic® Singeing Line by Bejimac Fig. 8. DORNIER singeing machine for circular knitted fabrics
- 17. What should control during singeing operation? Intensity and uniformity of flame Working speed of the fabric to be singed Effectiveness of singeing : should not add harshness in the fabric Should not damage the synthetic filament yarn in the fabric
- 18. Sizing /Slashing Slashing is the process where Size is applied to warp yarns for weaving. The purpose of size is to protect the yarn from the abrasive action of the loom. The process is carried out on a Slasher and the application procedure is called Sizing or Slashing.
- 19. NATURAL SIZING AGENTS Natural sizing agents are based on natural substances and their derivatives: Starch and starch derivatives; native starch, degradation starch and chemical modified starch products Cellulosic derivatives; carboxymethlycellulose (CMC), methylcellulose and oxyethlycellulose
- 20. SYNTHETIC SIZING AGENTS Polyacrylates Modified polyesters Polyvinyl alcohols(PVA)
- 21. DESIZING Desizing is the process of removing the size material from the warp yarns in woven fabrics. Sizing agents are selected on the basis of type of fabric,
- 22. Desizing Starch Once a starch solution dries, the resulting film will not readily re dissolve in water; therefore, to completely remove starch from a fabric, the polymer must be chemically degraded to make it water soluble. Three chemical methods can be used to degrade starch into water soluble compounds namely, Enzymes, Acid Hydrolysis and Oxidation.
- 23. Major Desizing Processes Rot steeping Enzymatic desizing Oxidative desizing Acid steepimg
- 24. Rot steeping Fabric is stored in warm water at 40-60o C overnight It remove the starch and water soluble impurities with natural reaction Drawback: Time consuming One can not certain that in the given duration size will remove completely
- 25. Oxidative steeping: Starch and other added impurities are hydrolysed through oxidation process followed by washing process. Oxidizing agents: Sodium Hypochlorite: 2.0-5.0 gm/lit. at room temperature at pH 7.0 Hydrogen peroxide:3.0-6.0 gm/lit of H2O2 and 7.0-15.0 gm/lit Sodium Hydroxide, 40oC for 12-16 hours Drawback: Time consuming Process Some time bleaching effect also obtain(If not require in the end product)
- 26. Acid steeping Acid hydrolysis lowers the molecular weight and reduce starch to glucose. Fabric is treated with Sulphuric Acid (0.5% to 1.0 %) at 40o C It also remove the starch and water soluble impurities with natural reaction Drawback: Tendering of cellulosic fibre if precautions are not taken If the acid remain in the fabric, whole lot may get seriously damage or tendered
- 27. Oxidative Desizing of strach The oxidative desizing methods are relatively unimportant and when using them, damage to fiber can never be completely avoided.
- 28. Enzymatic Desizing Enzyme solution in water with 0.5% to 2.0% on weight of fabric enzyme Require quantity of Common salt is also required Neutral pH Other method fabric is run continuously in machine having enzyme solution.
- 29. ENZYME Enzymes are high molecular weight protein biocatalyst that are very specific in their action. Enzymes are named after the compound they break down, for example: Amylase breaks down amylose and amylopectin, Maltase breaks down maltose and Cellulase breaks down cellulose. For desizing starch, amylase and maltase are used.
- 30. WORK OF ENZYMES Amylase will degrade starch into maltose, a water soluble disaccharide and Maltase will convert maltose into glucose, a simple sugar.
- 31. Quality control check Desizing efficiency test Iodine test Fluidity test
- 32. JIGGER A Jig or Jigger is a batch machine which handles fabric in open width. It is a simple machine consisting of let-off and take-up rolls, a trough containing the processing liquor and guide rolls which allow the fabric to pass from the let-off to the take-up roll.
- 33. Scouring It is process to remove all undesirable impurities (Natural: like dirt, vegetable matter, grease wax etc and Added: like remaining size material, stains etc.) Natural Impurities: Based on the composition of natural material like cotton, wool, silk etc.
- 34. SCOURING Natural fibers contain oils, fats, waxes, minerals, leafy matter and motes as impurities that interfere with dyeing and finishing. Synthetic fibers contain producer spin finishes, oils and/or knitting oils, grease. These impurities are not soluble in water, they can be removed by Extraction, dissolving the impurities in organic solvents, Emulsification, forming stable suspensions of the impurities in water and Saponification, Converting the contaminates into water soluble components.
- 35. Objectives To make the fabric highly hydrophillic. To remove impurities such as oil, wax, gum, husk as nearly as possible. To increase absorbency of fabric or textile material with out physical or chemical change. To produce a clean material by adding alkali to make the fabric ready for next process. To remove non cellulosic substances in case of cotton.
- 37. The change occurring of cotton fibres during scouring
- 38. Chemical used in scouring process
- 39. Form of scouring Yarn scouring : Hank Package Fabric scouring : open width Jigger Pad batch Rope form Kier
- 40. Methods of scouring There are two principal methods of scouring Discontinuous method (winch , jigger kier) Continuous method (scouring in j box )
- 41. J BOX
- 42. J Box Process
- 43. J box
- 44. Kier boiler
- 45. Difference between scouring and souring
- 46. SOLVENT SCOURING Certain organic solvents will readily dissolve oils fats and waxes and these solvents can be used to purify textiles. Removal of impurities by dissolution is called Extraction. There are commercial processes where textiles are cleaned with organic solvents. Fabrics processed this way are said to be "Dry Cleaned".
- 48. Bleaching This is a process of whitening-fibers, yarns, or fabrics having natural colour. It is carried out by using oxidising agents like Sodiumhypochlorite (NaOCl) Hydrogen peroxide (H2O2) Sodium Chlorite (NaClO2)
- 49. The color producing agents in natural fibers are often organic compounds containing conjugated double bonds. It is known in dye chemistry that conjugation is necessary for an organic molecule to perform as a dyestuff. Decoloration can occur by breaking up the chromophore, most likely destroying one or more of the double bonds within the conjugated system.
- 50. Bleaching Oxidative bleaches oxidize color bodies into colorless compounds. For example, double bonds are known to be oxidize into epoxides which easily hydrolyze into diols. The major bleaching agents used in textile preparation are sodium hypochlorite, hydrogen peroxide and sodium chlorite.
- 52. 52 Sodium Hypochlorite (NaOCl) Sodium hypochlorite is made by bubbling chlorine into a solution of sodium hydroxide. when acid is added to a hypochlorite solution, chlorine gas is liberated. HOCl is active bleaching agent (hypochlorous acid )
- 53. Antichlor Fabrics bleached with hypochlorite will develop a distinctive chlorine odor. This odor can easily be removed with an aftertreatment consisting of sodium bisulfite and acetic acid.
- 54. Souring After the sodium hypochlorite bleaching some CaCl2 , MgCl2 and other miniral matter live in fabric. After that the fabric feels harsh and stiff. To reduce these chlorides fabric treat with Hcl or sulphuric acid.
- 55. H202 Bleaching Hydrogen peroxide was first used to bleach cotton in the 1920's. By the invention of the J-box which lead to continuous processing. Today, it is estimated that 90 to 95 % of all cotton and cotton/synthetic blends are bleached with hydrogen peroxide.
- 56. Mechanism Hydrogen peroxide is a weak acid and ionizes in water to form a hydrogen ion and a perhydroxyl ion. The perhydroxyl ion is the active Hydrogen peroxide can also decompose. This reaction is catalyzed by metal ions e.g. Cu++, Fe+++. This reaction is not desired in bleaching because it is an ineffective use of hydrogen peroxide and causes fiber damage.
- 57. Effect of pH At pH < 10, hydrogen peroxide is the major specie so it is inactive as a bleach. pH 10.2 to 10.7 is optimum for controlled bleaching. Sodium hydroxide is used to obtain the proper pH. At pH > 11, there is a rapid generation of perhydroxyl ions. When the pH reaches 11.8, all of the hydrogen peroxide is converted to perhydroxyl ions and bleaching is out of control.
- 58. Stabilizers Stabilizers must be added to the bleach solution to control the decomposition of hydrogen peroxide. Stabilizers function by providing buffering action to control the pH at the optimum level and to complex with trace metals which catalyze the degradation of the fibers. Stabilizers include sodium silicate, organic compounds and phosphates.
- 59. Sodium Silicates Sodium silicates are the most commonly used and most effective hydrogen peroxide bleach stabilizers. silicates have a natural affinity for ferrous ions and ferrous ions are naturally present in cotton. The silicates are adsorbed onto the ferrous ions in the fiber, producing a species that catalytically enhances bleaching while reducing bleach decomposition and fiber damage.
- 60. Test For Degree of Bleaching Whiteness Index-CCM
- 61. Whiteness The standard ceramic tile is measured and set to equal 100. The other specimen are rated against this standard. Unbleached fabrics will give values in the 50 to 60 range. Well breached fabrics will rank 95 or better.
- 62. OPTICAL BRIGHTENERS Certain organic compounds possess the property of fluorescence which means that they can absorb shorter wave-length light and re-emit it at longer wave-lengths. A substance can adsorb invisible ultra-violet rays and re- emit them within the visible spectrum. Therefore a surface containing a fluorescent compound can emit more than the total amount of daylight that falls on it, giving an intensely brilliant white. Compounds that possess these properties are called Optical Brighteners or OBA's.
- 63. OBA If brilliant white finish is required in textile, then bleaching is not sufficient. For this purpose Fluorescent brightening agents are used. For synthetic fibres-OBA –Optical Brightening Agents are mostly used.
- 64. • Fluorescence is the ability of a substance to absorb radiation of light and re-radiate it at a longer wavelength. Incident light Visible U.V Fibre without FBA Fibre with FBA Eye detect visible only Reflected UV & Visible Eye detect visible & UV both Reflected Visible Plus UV converted to visible Incident light Fluorescent brightening agent(FBA)
- 65. OBA /FBA /BA Optical brightening agents are based on (DAST) diamino stilebenedisulfonic acid Oba absorb ultraviolet light to within the range of 350 to 360nm and reemit the absorbed energy at 400 t0 500 nm with a maximum wavelength at 430nm. Due to presence of sulphonic acid group present in OBA structure contribute to water solubility.
- 66. Properties of OBA Mostly they are anionic in charge (negetive ) pH typically between 8.5 to 11.0. OBA addition will impart Brightness Whiteness b* value(+b-yellow,-b blue ) Lvalue a* value (lower green), (-a red)
- 67. Types of OBA Basic classes of chemicals used in OBAs include: Triazine-stilbenes (di-, tetra- or hexa- sulfonated) Disulphonated (cellulose) Tetrasulphonated (universal) Hexasulphonated Coumarins Imidazolines
- 68. 68 The Examples of preparation processes of cotton and its blends fabrics 100% Cotton fabrics 1. Singeing 2. Desizing (only for woven fabrics) 3. Scouring 4. Bleaching (only for pale or bright shades) 5. Mercerizing 6. Adding of Optical Brightening Agent
- 69. 69 100% Silk Fibers 1. Degumming (to get rid of non-fibrous materials (sericin)) 2. Bleaching (to make the goods whiter)
- 70. 70 100% Wool Fibers 1. Carbonising (to remove leave, bark and other parts of the plants from the wool fibers) 2. Scouring (to get rid of fat and oil from the fibers) 3. Bleaching (to make the fibers whiter)
- 71. Scouring of Wool • Wool is a protein fibre and sensitive to bases. Wool can be scoured by using detergent at pH upto 7 at 60o C • To remove vegetable material it is carbonised by using Dilute Sulphuric acid. • It has tendency of felting
- 72. Carbonising of wool • Carbonising is a chemical treatment of wool with strong acid (H2SO4)designed to remove any remaining vegetable impurity.
- 73. Felting and milling • Felting is the tendency of the fibres to form matted entanglements such that a woven fabric, for example, can become so matted that the warp and weft yarns are no longer visible. • The result is a fabric which is apparently composed entirely of a entangled web of fibres and the fabric is referred to a felt. • Milling is the process of deliberately felting wool goods to achieve the effects of felting.
- 74. Natural Impurities-Silk • Silk contain 22 to 25% gum
- 75. Scouring of Silk De-gumming: • It is scoured by using Soap and soda ash • Treatment for 1 hour at 50o C (repeat this process three times) • Bleaching is carried out with hydrogen peroxide (No NaOH) With sodium silicate
- 76. Mercerization This is the process applicable only on cellulosic fibres especially cotton. The main purpose of mercerization is to alter the chemical and physical properties of the fibre.
- 77. Change in cross section 1.Cross section of cotton before mercerisation 2-5 swelling process in 18% NaOH 66 Rinsing process after swelling 7 Final state Change in cross section
- 78. Mercirization /Causticization Both Mercerization and causticization require cotton to be treated with concentrated solutions of sodium hydroxide (caustic soda). Mercerization requires higher concentrations of caustic soda (19 to 26 % solutions) whereas causticization is done with concentrations ranging between 10 to 16%. One major difference between the two is that causticizing improves the dyeing uniformity and dye affinity of cotton without improving luster.
- 79. Mercirization / Causticization Caustic soda solution swells cotton fibers breaking hydrogen bonds and weak van der Waal forces between cellulose chains. The expanded, chains rearrange and re-orient and when the caustic soda is removed , the chains form new bonds in the reorganized state. When done tensionless, the cotton fiber swells, the cross section becomes thicker and the length is shortened. Because of fiber thickening, the fabric becomes denser, stronger and more elastic. Under tension, The fibers become permanently round and rod like in cross section and the fiber surface is smoother, adding to fiber luster.
- 80. Objectives improved luster increased ability to absorb dye improved reactions with a variety of chemicals improved strength/elongation improved smoothness improved hand
- 81. Procedure If cotton is dipped into a strong alkaline solution such as lithium hydroxide, caustic soda, or potassium hydroxide,The fibers will swell and shrink. If the fibers are placed under tension while in this swollen state and then rinsed with water, the alkali will be removed and a permanent silk-like luster will result.
- 82. Effect of alkali on cotton fibres
- 83. PHYSICAL CHANGE Improved in strength Improved in lusture (under tension) Change in cross section from bean shape to almost circular change in longitudinal view convoluted to cylinderical
- 84. Chemical change Improvement in moisture regain improvement in dye uptake Formation of celluloseII
- 86. Mercerising can be carried out on yarn in hanks, woven and knitted fabric through one of the following different treatments: · mercerising with tension · caustification (without tension) · ammonia mercerising.
- 87. Ammonia mercerising Cotton yarn and fabric can be treated with anhydrous liquid ammonia as an alternative to caustic soda. Effects similar to mercerising are obtained, although the lustre grade is inferior to caustic soda mercerising. Traces of ammonia have to be removed, preferably with dry heat treatment followed by steaming. This method is not widely used. Only a few plants are reported to use ammonia mercerising in Europe.
- 88. Mercerization methods Tension Mercerization (treatment under tension) Slack Mercerization (treatment without tension)
- 89. Mercerization machines Yarn mercerization Fabric mercerization Fabric mercerization: Pad chain type mercerization Pad chainless type mercerization Padless chainless type mercerization
- 90. Test for Mercerization Barium Activity Number AATCC Test Method 89 is a common test used for quantifying the degree of Mercerization. It is based on the fabric‘s ability to absorb barium hydroxide. The procedure is carried out on the fabric both before and after Mercerizing and the barium number is calculated as shown below.
- 91. Test for Mercerization Unmercerized fabric will give a barium number of 100 to 105. Completely mercerized fabric will give a barium number of 150. Commercially treated fabrics fall in a range between 115 to 130.
- 92. Yarns are Mercerized as continuous sheets in a fashion similar to woven fabrics. Greige yarns enter the caustic saturator and are held so they do not shrink until the caustic has been washed out. Afterwards, the yarns are dried continuously and wound onto bobbins or spools. These conditions will develop satisfactory luster, elasticity and dyeing properties; however, if higher luster and strength is desired, the yarns should be stretched beyond the greige length during the process. Increase stretch, however, reduces dye affinity and elasticity. The reasons for mercerizing yarns are: 1. To produce lustrous, strong 100 % cotton sewing thread. 2. To produce yarns for constructions that are difficult to mercerize. 3. To produce yarns for special construction effects. 4. To produce yarns for blends containing fibers too sensitive formercerizing.
- 93. Work Procedure In natural condition the cotton fiber is a flat, twisted, ribbon-like filament. When immersed in caustic solution it swells out and takes on a round and a hair like appearance, and becomes plump instead of flat. How it is done The cotton is soaked in strong caustic soda or caustic potash solution for a few moments under stretch and then washing in pure water to remove the caustic. trong caustic soda or caustic potash solution for a few moments under stretch and then washing in pure wat
- 94. The cellulose is changed into hydro- cellulose or cellulose-hydrate. Cellulose cannot be dyed so easily. Hydro- cellulose on the other hand, absorbs almost any kind of dye readily. Mercerised cotton takes dyes so fast, that chemicals are added in the dye bath to check the process in order that the dyes may not enter so rapidly as to render the shading uneven.
- 95. Woven fabric
- 96. Knitted
- 97. Taking a cotton blend, and then mercerising will produce an effect called as crepon effect. Similarly, mercerisation can be used to produce seersucker effect by Mercerising only certain stripes on the warp direction by covering the rest of the cloth by suitable means.
