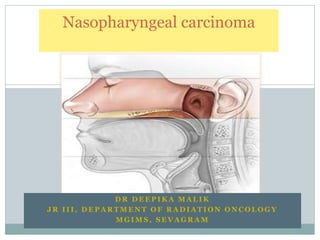
Nasopharyngeal cancer
- 1. D R D E E P I K A M A L I K J R I I I , D E P A R T M E N T O F R A D I A T I O N O N C O L O G Y M G I M S , S E V A G R A M Nasopharyngeal carcinoma
- 5. Lateral wall •Contain the eustachian tube •Torus tubarius- elevation of mucous membrane of lateral nasopharynx formed by cartilage of eustachian tube •Fossa of rosenmuller-* pharyngeal recess lying posterior to torus
- 7. Posterior wall Superior pharyngeal constrictor muscle, pharygobasillar fascia, buccopharyngeal fascia Superior constrictor extends to base skull only in midline Laterally , Pharyngobasillar fascia attaches it to base skull at basiocciput and petrous part of temporal bone
- 8. Sinus of morgagni- muscular deficiency Eustachian tube and levator veli palatini pass through it Significance ?
- 10. Jugular foramen Base of skull Foramen magnum Foramen ovale Foramen spinosum Foramen lacerum Stylomastoid foramen Hypoglossal canal
- 12. Epidemiology Uncommon Overall incidence world- 0.6/ 1 lakh population India – 17.2 per 1 lakh people per year Highest incidence- South China. ( 17.8 in Hong Kong; 26.9 in Guangdong province) china
- 13. Age- in low risk populations- bimodal age distribution ( 15- 25 years) (50-59 years) In high risk populations- 4th and 5th decade Sex- similar age distribution M:F =2:1 to 3:1
- 14. Etiological features Genetics- -High incidence in southern China and descendants of south China -Genome study- - HLA haplotypes- A2, B46, B17 – high risk •Environment -Salted fish in Southern China ( dimethyl nitrosamine- carcinogen) -alcohol -exposure to dust, fumes, formaldehyde -exposure to cigarette smoke •Epstein – barr virus •Tumorogenic potential – set of latent genes : --- -latent membrane proteins (LMP1, LMP2A, LMP2B) - EBV- determined nuclear antigens (EBNA1 , EBNA2) •LMP1- principal oncogene, 80-90% of NPC
- 15. Local extension- anteriorly Nasal fossae destruction of lateral wall destruction of pterygoid plate Advanced cases- infiltration of orbital apex ( through IOF)
- 16. Local extension- inferiorly Oropharynx Advanced disease- invasion of C1
- 17. Local extension- lateral Lateral parapharygeal space, invasion of levator and tensor veli palatini muscle
- 18. Local extension- superior and posterior Base of skull, sphenoid sinus, clivus
- 20. Lateral to the pharyngobasilar fascia, the nasopharynx is bounded by four spaces These include the masticator (infratemporal fossa), the parapharyngeal, the carotid and the parotid space
- 21. Lymphatic spread Vast avalvular lymphatic network in mucous membrane frequent involvement of regional lymph nodes. -85-90% cases present with ipsilateral nodes -Approxmately 50 % present with bilateral nodes
- 22. Upper jugular-94% Middle juular-85% Retropharyngeal node- 80% Posterior cervical -46% Lower jugular-19% Supraclavicular -17% Submental-17%
- 23. 2 major lymph collectors 1. Lateral side of NPx lateral pharyngeal nodes, jugulodigstric, and 3rd, 4th , 5th retropharyngeal group 2. Posterior collector 1st group of RPN ( node of rouviere) Further metastatic spread to midjugular, lower jugular , posterior cervical , supraclavicular nodes
- 24. Haematogenous spread 3-6% cases- distant metastasis at presentation 18-50 % cases- distant metastasis during disease course bone > lungs and liver ( lung mets- better prognosis)
- 25. Clinical presentation Neck mass Most common presentation ( 66 %) u/l, b/l Typically: mass in upper posterior neck , beneath superior portion of SCM close to mastoid process •Nasopharyngeal mass •37% •Epistaxis,,Nasal obstruction, Nasal discharge •Base of skull extension Headache – 40 % Cranial nerve involvement- 23 %
- 26. Syndromes associated with NPC Cranial Nerve compression II to VI ( direct extension intracranially) as they emerge from cranial vault at or near base of skull orifices M/c - cranial nerve V and VI
- 27. Retroparotid Syndrome of VILLARET Enlarged lateral retropharyngeal lymph metastasizing to retroparotid space. Involves IX to XII cranial nerve & Cervical Sympathetic trunk. Difficulty in swallowing ( IX, X) Perversion of taste in posterior 3rd of tongue( IX) Hyper or hypoaesthesia of mucous membrane of soft palate , pharynx, larynx (X) Hemiparesis of soft palate , paralysis and atrophy of trapezius and SCM (XI) Unilateral paralysis and atrophy of tongue (XII) Horner syndrome (cervical sympathetic chain)
- 28. Horner syndrome
- 29. PETROSPHENOID SYNDROME of JACOD II- VI CN Unilateral neuralgia of V nerve Unilateral ptosis (III) Complete ophthalmoplegia (III,IV,V) Amaurosis (II)
- 30. TROTTER’S TRIAD/ SOM syndrome NPC Neuralgia I/L ( V) Conductive hearing loss (VIII) Palatal Palsy (X)
- 31. Diagnosis is made by Biopsy Local anaesthesia (OPD) G.A. ( deep tumour, uncooperative patient) At times- tumour invisible, submucosal For suspicious NPC cases- random biopsies of most commonly involved sites b/l fossa of rosenmuller superior posterior wall FNAC of suspicious neck mass- to establish metastasis in regional lymph nodes
- 32. Physical examination Palpation of neck node (size, laterality, lowest extent of largest node, supraclavicular fossa inv) Cranial nerve exam ( vision and hearing) Chest percussion and ausculation Abdominal palpation Spine and bone exam Nasopharyngoscopy and biopsies Panendoscopy +/-
- 33. Lab studies CBC LFT EBV specific serological tests - Ig A - VCA (Ig A antiviral capsid antigen) - antiviral capsule antigen titres - serum EBV DNA levels (prognosis, surviellance post treament )
- 34. Imaging for locoregional extent MRI- study of choice Why? AJCC staging requires a search for invasion into soft tissue ( parapharyngeal space), bony structures. MRI is superior to CT in delineating muscle, soft tissue and skull base. Thin slices (3 mm). ( thicker slices, >5mm , risk misdiagnosis)
- 35. Ng et al : compared MRI and CT in assessing disease extent. Higher sensitivity of MRI for skull base inv (60% vs 40%), intracranial inv (57% vs 36%), retropharyngeal node( 58%vs 21 %), prevertebral muscle infiltration ( 51% vs 22%) MRI modified staging in 27% ( 23% upstaged, 4% downstaged)
- 36. Imaging for nodal metastasis MRI , CT LN metastasis radiologically defined as ( Van den Brekel) -presence of central necrosis -extracapsular spread -SAD>= 10 mm ( 11mm for jugulodigastric , 5mm retropharyngral node) -cluster of 3 or more LN that are borderline in size.
- 37. Imaging for metastatic workup If clinically indicated or N3 disease PET-CT :study of choice (Sensitivity-100%, specificity-90%) Others- CT chest( clinical suspicion of lung met) CT abdomen ( abnormal LFT, clinical) Bone scan- clinical suspicion, raised Alk Ph
- 38. Staging systems AJCC identical UICC Ho system : advantage- N stage classification ( level or location)
- 39. Comparison System Staging T1 T2 T3 T4 Fletcher (1967) < 1 cm diameter > 1 cm but confined to nasopharynx Beyond nasopharynx Involving skull base or cranial nerves Ho (1978) Confined to nasopharynx Extending to nasal fossa or oropharynx Bone/ Cranial nerve/ orbital / hypopharyngeal / infratemporal fossa involvement NA IUAC (1988) Limited to one site in nasopharynx Extending to two sites in nasopharynx No bony destruction Bony destruction including eustachian tube Huaqing (1994) Limited to nasopharynx Involving the nasal cavity, oropharynx, anterior cervical vertebrae, PPS before SO line Pterygoid process / posterior cranial nerve / posterior cervical vertebrae / BOS / PPS beyond SO line Infratemporal fossa / cavernous sinus / PNS / direct invasion of C2 or C1 / anterior cranial nerves
- 40. AJCC 2010 Tx T0 Tis T1-tumor confined to NPhx or extends to nasal cavity and/or oropharynx (soft palate, C1/C2) without parapharygeal exetension T2- with parapharyngeal extension (beyond Pharyngobasilar fascia) T3-bony structures of skull base and/or paranasal sinuses T4- with intracranial extension and/or involvement of cranial nerves, hypopharynx, orbit, infratemporal fossa, masticator space ( beyond anterior surface of lateral pterygoid muscle, or lateral extension beyond posterolateral wallof maxillary antrum and pterygomaxillary fissure) HPx, orbit, masticator spce
- 41. Nx N0 N1- U/L cervical LN above supraclavicular fossa </= 6 cm OR U/L or B/L retropharyngeal LN </= 6 cm N2- B/L cervical LN above supraclavicular fossa </= 6 cm N3a – LN > 6cm N3b- LN below SCF U/L neck node ( above SCF) or B/L retopharyngeal nodes <6cm
- 42. Pathologic classification 80-99 % - carcinomas ~ 5%- lymphomas ( adenocarcinoma, plasmacytoma, melanoma, sarcoma)
- 43. WHO 2005 classification- NPC 1.Keratinising SCC- keratin pearls 2.Non keratinising differentiated SCC 2.Non keratiniing undiff SCC To 4.Basaloid SCC ( palisading)
- 44. WHO 2005 classification- NPC Keratinising SCC- keratin pearls Non keratinising differentiated SCC Non keratiniing undiff SCC To Basaloid SCC Non keratinising type- strong association with EBV Lymphoepithelioma/ lymphoepithelial carcinoma- variant of undifferentiated SCC
- 45. Treatment
- 47. Radiation therapy Primary treatment After 1950, surgical interventon rare
- 48. Positioning: Supine position, head extended Immobilization a custom-made thermoplastic cast covering head to shoulder region
- 49. Dose 70 Gray / 7 weeks , 50-60 Gray to potential risk sites Local control significantly improved in patients receiving > 67 Gy to tumor target. Marks et al, Vikram et al Perez etal •T1-T2 , (local control rate of 100%, >70 Gy ) Vs (80 %, 66-70 Gy) • T3-T4 , local control <55%, even with >70 Gy Mesic etal – T3-4 tumors, doses >60 Gy or larger portals, no improvement on outcome
- 50. Fraction 2 Gray/ fraction •Lee et al •T1 , 4 fractionation schedules •Total dose was important factor •Dose / fraction did not affect local control •Dose /fraction was a signifcant risk factor for temporal lobe necrosis
- 51. Time No interruptions •Marcial etal •Split course irradiation (30 Gy/10 Fr/ 2 weeks --- 3 week rest--- 30 Gy/10 Fr/ 2 weeks Vs •66 Gy/33 Fr/6.5 to 7 weeks • similar 5 year local control and DFS Vikram etal • interruption >21 days poorer local control •Many subsequent studies show similar results
- 52. Volumes Nasopharynx , adjacent parapharyngeal tissue ( 1-2 cm margin) and cervical lymphnodes. Also include posterior ethmoid cells, post 1/3rd maxillary antrum and nasl cavity
- 53. Conventional 2 D technique ( chao, perez, brady) Opposing lateral portals ( tumor + upper nodes) AND matching lower anterior cervical field for lower neck nodes After 45 Gy, shield spinal cord
- 54. GTV- nasopharyngeal tumor , gross retropharyngeal lymphadenopathy, gross nodal disease. N0 disease- prophylactic irradiation recommended ( high incidence of occult metastasis) CTV: GTV +regions of microscopic disease Definition of margins and dose levels may be different in different centres
- 55. CTV 70 CTV 59.4 (High risk subclinical) ( all potential areas of microscopic spread) PTV GTV + >/= 5 mm margin ( can be reduced to 1mm for tumors close to critical structure, brainstem, SC CTV70 + >/= 5 mm margin •Entire nasopharynx •Retropharyngeal LN •Clivus •Base skull •Pterygoid fossae •Parapharyngeal space •Sphenoid sinus •Posterior ¼th to 1/3rd of nasal cavity •Posterior 1/4th to 1/3rd of maxillary sinuses •High risk nodal levels ( all bilaterally) •{ upper deep jugular, level I, level II, level III, level IV, level V, retropharyngeal } •CTV + circumferential margin of 3-5 mm to all CTV’s ( margin may be decreased to 1mm , close to critical structures)
- 56. Note- CTV margins- mat be limited to exclude bone not at risk for subclinical disease or air. Bilateral IB LN can be spared if patient is node negative *
- 57. IMRT – normal tissue dose constraints Brain stem <5Gy Optic N <54 Gy Optic chiasm <54 Gy SC <45 GY or 1 cc of PTV cannot exceed 50 Gy Mandible , TM Jt <70 Gy 75 Gy Brachial plexus <66 Gy Temporal lobe <60 Gy Oral cavity 40 Gy Parotid <26 Gy Eyes <35 Gy Lens <25 Gy Larynx <45 Gy 1 % of PTV cannot exceed 60 Gy
- 58. 3 D CRT Memmorial Sloan Kettering cancer centre, new york---- 3D planning vs 2 D. better dose coverage to tumor, decreasing issues Liebel et al- • mean tumor dose increased by 13% •Tumor control increased by 15 % •Jen et al •Significant improvement in 3 year L-FFR for T4 (86% vs 47 •Significant improvement in in EFS for stage III and IV •Incidence of xerostomia – significantly less
- 59. IMRT is replacing conventional RT * Overstringent use of normal tissue constraints – inadequate coverage of tumot targets** UCSF • First • 70 Gy , PTV gross and inv LN ( 2.12-2.25 Gy/Fr • 59.4 Gy, PTV high risk subclinical( 1.8 Gy/Fr • 54 Gy , PTV low risk subclinical (1.64 Gy/Fr • Once daily • L-FFR- 96%, D-FFR- 72%, Nodal FFR- 98%
- 60. SMART ( dose painting) •MSKCC •AF by concommitant boost vs SMART •No significant improvement in 3 yr L- FFR •T3, T4 tumors, SMART •Locoregional control excellent •Serious late toxicities •4% - carotid artery pseudoaneurysm, haemmorhhage •4% - temporal lobe necrosis
- 61. 2 IMRT approaches 1. extended whole field IMRT 2. split field Which is better? – controversial *
- 62. Brachytherapy Dose escalation Intracavitary or interstitial implants T1-T3 NPC as a boost OR recurrent disease Not suitable when intracranial extension* ( bone inv) Presently its use is declining** HDR
- 63. Rotterdam applicator, others (Mould technique, Levendag’s Forzhou (Chinese district),Simple catheter based •Designed by Levendag • can be worn by the patient comfortably •Made up of silicone: flexible and closely conforms to the curvature of the nasopharynx. •A silicone bridge and flange used to fix the applicator
- 65. Prescription points Tumor points: Na (Nasopharynx) – 2 BOS (Base of Skull) - 2 R (Node of Rouviere) - 1 Normal Tissue points: OC ( Optic Chiasm) - 1 P (Pituitary gland) - 1 C (Cord) – 1 Pa (Soft Palate) – 2 Re (Retina) - 2 No ( Nose) - 2
- 66. SRS Dose escalation •T1-T4 •SRT boost 12 Gy following EBRT 66 Gy •OS- 69%, distant failure rate- 32% •12.1 % - temporal lobe necrosis •3.6% - retinopathy
- 67. Chemotherapy NPC – highly chemosensitive Stage I : RT Stage III-IVB : CTRT +/- adjuvant chemo Intermediate stages- lesser evidence, CTRT
- 68. Concurrent Chemoradiation Intergroup 0099 trial RT vs CRT * ( cisplatin) Significant OS benefit for CRT (78% vs 47%) at 3 years. Metaanalysis of 1o trials In favour of CRT
- 69. Adjuvant chemotherapy US intergroup regimen - Cisplatin and 5 FU Efficacy?* RCT’s- RT vs RT+ adj CT -- negative results RCT- CRT vs CRT+ adj CT- statistically not significant therefore,; optional therapy ** Neoadjuvant chemotherapy •RCT ph II- Docetaxel/cisplatin followed by CTRT vs CTRT OS benefit • •Ph III trial, taiwan - ICT*** CRT , excellent FFLF, FFDF, OS with acceptable toxicities ( ASTRO 2012) •Ongoing Ph III trial ( HongKong) , 3 arms#( cisplatin/5FU+CTRT vs CTRT+adj cisplatin/5FU vs cisplatin/capecitabine +CTRT -Estimated enrolment completion by april 2017
- 70. Persistent/ Recurrent NPC Persistent NPC- does not completely regress following primary treatment Recurrent NPC- reemerge after initial complete regression Persistent disease- better survival and control rates
- 71. When to consider it as persistent disease and proceed with t/t Perez etal recommend : observation period of 10 weeks before additional t/t * Bx Positive biopsies beyond 12 weeks indicate poor prognosis. •Early detection – crucial •Nasopharyngoscopy – more sensitive •Biosy – to confirm •MRI- delineate tumor extent, ( PNI, intracranial extension); superior to CT •FDG PET- superior to MRI ( Sn 100%, sp 93%) •Circulating EBV DNA- detect failure **
- 72. Additional RT for persistent disease Re –irradiation for recurrent disease Brachytherapy after a full course EBRT : 87-95% 5 year L-FFR (T1) SRT SRT 15 Gy vs HDR brachy 20 Gy ( T1-T4) 3 yr LFFR( 86% vs 71%) Brachytherapy Brachy + EBRT(better) 2D, 3D, IMRT (100% salavge rate, no complications)* SRS/SRT - to be avoided when carotid encasement- fatal Haemorrhage Concurrent chemo- Cisplatin Induction chemo- gemcitabine, cisplatin
- 73. Surgical treatment Persistent / recurrent nodal disease- RND If extension beyond nodes- additional brachy Approaches- infratemporal (L), transpalatal, transmaxillary, transcervical (I), anterolateral Recent- transnasal, endoscopic
- 74. Sequelae of treatment •Temporal lobe necrosis- most troublesome •65% of irradiation induced deaths in NPC •Larger fraction size>2 Gy •Difficult and delayed diagnosis* •Cranial neuropathy •m/c XII •( IX, X, XI, VI) •Slurring, dysphagia, aspiration •Oral complications •Xerostomia Parotid sparing IMRT, MPD- 34 Gy only after ruling out parotis inv •Osteoradionecrosis Px flouride, decayed teeth removal before RT Hearing loss • SNHL -More with cisplatin CTRT Mean cochlea dose < 48 Gy -Otitis media- eustachian tube damage Carotid artery injury Ruptured pseudoaneurysm Endocrine dysfunction Galactorrhoea, hyperprolactinaemia 2nd malignancy •Rare, 0.04%, after 10 yrs •Maxillary osteosarcoma Soft tissue sarcoma
