Exploring Intensified Seed Train Through Advancements in Perfusion Processing Technologies
•
0 likes•52 views
This poster explores key elements of bioreactor design and automation strategies that enable successful implementation of seed train intensification via perfusion: - Sparger performance characterization - Cell retention device connection - Evaluation of the Hamiltion® Incyte viable cell density (or permittivity) sensor - Cell culture case studies To learn more about this topic or collaborate with our technical experts, schedule an in-person or remote visit at our M Lab™ Collaboration Centers: www.emdmillipore.com/mlab
Report
Share
Report
Share
Download to read offline
Recommended
Recommended
❤️Chandigarh Escorts Service☎️9815457724☎️ Call Girl service in Chandigarh☎️ Chandigarh Call Girls Service ☎️ Call Girls In Chandigarh BEST Call Girls in CHANDIGARH Escort Service provide Cute Nice sweet and Sexy Models in beautiful CHANDIGARH city cash in hand to hand call girl in CHANDIGARH and CHANDIGARH escorts. HOT & SEXY MODELS // COLLEGE GIRLS IN CHANDIGARH AVAILABLE FOR COMPLETE ENJOYMENT WITH HIGH PROFILE INDIAN MODEL AVAILABLE HOTEL & HOME ★ SAFE AND SECURE HIGH CLASS SERVICE AFFORDABLE RATE ★ 100% SATISFACTION,UNLIMITED ENJOYMENT. ★ All Meetings are confidential and no information is provided to any one at any cost.
★ EXCLUSIVE Profiles Are Safe and Consensual with Most Limits Respected
★ Service Available In: - HOME & 24x7 :: 3 * 5 *7 *Star Hotel Service .In Call & Out call
Services :
★ A-Level (5 star escort)
★ Strip-tease
★ BBBJ (Bareback Blowjob)Receive advanced sexual techniques in different mode make their life more pleasurable.
★ Spending time in hotel rooms
★ BJ (Blowjob Without a Condom)
★ Completion (Oral to completion)
★ Covered (Covered blowjob Without a Condom)Chandigarh Call Girls❤️Chandigarh Escorts Service☎️9815457724☎️ Call Girl service in Chandigarh☎️ ...

❤️Chandigarh Escorts Service☎️9815457724☎️ Call Girl service in Chandigarh☎️ ...Rashmi Entertainment
More Related Content
More from MilliporeSigma
More from MilliporeSigma (20)
Moving your Gene Therapy from R&D to IND: How to navigate the Regulatory Land...

Moving your Gene Therapy from R&D to IND: How to navigate the Regulatory Land...
Identity testing by NGS as a means of risk mitigation for viral gene therapies

Identity testing by NGS as a means of risk mitigation for viral gene therapies
Latest advancements of melt based 3D printing technologies for oral drug deli...

Latest advancements of melt based 3D printing technologies for oral drug deli...
CAR-T Manufacturing Innovations that Work - Automating Low Volume Processes a...

CAR-T Manufacturing Innovations that Work - Automating Low Volume Processes a...
How does the ICH Q5A revision impact viral safety strategies for biologics?

How does the ICH Q5A revision impact viral safety strategies for biologics?
Improve Operational Efficiency by Over 30% with Product, Process, & Systems A...

Improve Operational Efficiency by Over 30% with Product, Process, & Systems A...
Insights from a Global Collaboration Accelerating Vaccine Development with an...

Insights from a Global Collaboration Accelerating Vaccine Development with an...
Risk-Based Qualification of X-Ray Sterilization for Single-Use Systems

Risk-Based Qualification of X-Ray Sterilization for Single-Use Systems
Rapid Replication Competent Adenovirus (rRCA) Detection: Accelerate your Lot ...

Rapid Replication Competent Adenovirus (rRCA) Detection: Accelerate your Lot ...
The High Intensity Sweeteners Neotame and Sucralose: 2 Ways to ace the Patien...

The High Intensity Sweeteners Neotame and Sucralose: 2 Ways to ace the Patien...
The Developability Classification System (DCS): Enabling an Optimized Approac...

The Developability Classification System (DCS): Enabling an Optimized Approac...
EU GMP Annex 1 Draft - Closed System Design Consideration with Single-Use Sys...

EU GMP Annex 1 Draft - Closed System Design Consideration with Single-Use Sys...
Introducing our novel Sf9 rhabdovirus-negative (Sf-RVN®) Platform

Introducing our novel Sf9 rhabdovirus-negative (Sf-RVN®) Platform
The Viscosity Reduction Platform: Viscosity-Reducing Excipients for Protein F...

The Viscosity Reduction Platform: Viscosity-Reducing Excipients for Protein F...
Addressing Downstream Challenges with Complex Injectables

Addressing Downstream Challenges with Complex Injectables
The Viscosity Reduction Platform: Enabling subcutaneous (subQ) delivery

The Viscosity Reduction Platform: Enabling subcutaneous (subQ) delivery
Webinar: How a Helium Manufacturing Release Test Increases Integrity Assuranc...

Webinar: How a Helium Manufacturing Release Test Increases Integrity Assuranc...
Webinar: Effective and Efficient Design of a Downstream Purification Process ...

Webinar: Effective and Efficient Design of a Downstream Purification Process ...
Endotoxin Control and Clearance in Biomanufacturing

Endotoxin Control and Clearance in Biomanufacturing
Recently uploaded
❤️Chandigarh Escorts Service☎️9815457724☎️ Call Girl service in Chandigarh☎️ Chandigarh Call Girls Service ☎️ Call Girls In Chandigarh BEST Call Girls in CHANDIGARH Escort Service provide Cute Nice sweet and Sexy Models in beautiful CHANDIGARH city cash in hand to hand call girl in CHANDIGARH and CHANDIGARH escorts. HOT & SEXY MODELS // COLLEGE GIRLS IN CHANDIGARH AVAILABLE FOR COMPLETE ENJOYMENT WITH HIGH PROFILE INDIAN MODEL AVAILABLE HOTEL & HOME ★ SAFE AND SECURE HIGH CLASS SERVICE AFFORDABLE RATE ★ 100% SATISFACTION,UNLIMITED ENJOYMENT. ★ All Meetings are confidential and no information is provided to any one at any cost.
★ EXCLUSIVE Profiles Are Safe and Consensual with Most Limits Respected
★ Service Available In: - HOME & 24x7 :: 3 * 5 *7 *Star Hotel Service .In Call & Out call
Services :
★ A-Level (5 star escort)
★ Strip-tease
★ BBBJ (Bareback Blowjob)Receive advanced sexual techniques in different mode make their life more pleasurable.
★ Spending time in hotel rooms
★ BJ (Blowjob Without a Condom)
★ Completion (Oral to completion)
★ Covered (Covered blowjob Without a Condom)Chandigarh Call Girls❤️Chandigarh Escorts Service☎️9815457724☎️ Call Girl service in Chandigarh☎️ ...

❤️Chandigarh Escorts Service☎️9815457724☎️ Call Girl service in Chandigarh☎️ ...Rashmi Entertainment
🍑👄Ludhiana Escorts Service☎️98157-77685🍑👄 Call Girl service in Ludhiana☎️Ludhiana Call Girls Service 🍑👄 Call Girls In Ludhiana Book Now :- 98157-77685
Our agency presents a selection of young, charming call girls available for bookings at Oyo Hotels. Experience high-class escort services at pocket-friendly rates, with our female escorts exuding both beauty and a delightful personality, ready to meet your desires. Whether it's Housewives, College girls, Russian girls, Muslim girls, or any other preference, we offer a diverse range of options to cater to your tastes.
We provide both in-call and out-call services for your convenience. Our in-call location in Delhi ensures cleanliness, hygiene, and 100% safety, while our out-call services offer doorstep delivery for added ease.
We value your time and money, hence we kindly request pic collectors, time-passers, and bargain hunters to refrain from contacting us. l Ludhiana, Majestic Grand Hotel, Ramada by Wyndham Ludhiana City Centre, Park Plaza Ludhiana, Windsor Fountain, G.T Road Ludhiana escort all Ludhiana service Russian available model female girls in Ludhiana VIP Lo price personal Ludhiana off class call girls payment high profile model and female escort 70% Off On Your First Booking Ludhiana Call Girls Service Cash Payment
Welcome to DILPREET Ludhiana Call Girl Service, the Trusted call girl agency around. We Offer 70% Discount On Your First Booking For Ludhiana Call Girls Service Cash Payment is available.🍑👄Ludhiana Escorts Service☎️98157-77685🍑👄 Call Girl service in Ludhiana☎️Ludh...

🍑👄Ludhiana Escorts Service☎️98157-77685🍑👄 Call Girl service in Ludhiana☎️Ludh...dilpreetentertainmen
❤️Amritsar Call Girls Service☎️98151-129OO☎️ Call Girl service in Amritsar☎️ Amritsar Call Girls Service ☎️ Call Girls In Amritsar BEST Call Girls in Amritsar Escort Service provide Cute Nice sweet and Sexy Models in beautiful Amritsar city cash in hand to hand call girl in Amritsar and Amritsar escorts. HOT & SEXY MODELS // COLLEGE GIRLS IN Amritsar AVAILABLE FOR COMPLETE ENJOYMENT WITH HIGH PROFILE INDIAN MODEL AVAILABLE HOTEL & HOME ★ SAFE AND SECURE HIGH CLASS SERVICE AFFORDABLE RATE ★ 100% SATISFACTION,UNLIMITED ENJOYMENT. ★ All Meetings are confidential and no information is provided to any one at any cost.
★ EXCLUSIVE Profiles Are Safe and Consensual with Most Limits Respected
★ Service Available In: - HOME & 24x7 :: 3 * 5 *7 *Star Hotel Service .In Call & Out call
Services :
★ A-Level (5 star escort)
★ Strip-tease
★ BBBJ (Bareback Blowjob)Receive advanced sexual techniques in different mode make their life more pleasurable.
★ Spending time in hotel rooms
★ BJ (Blowjob Without a Condom)
★ Completion (Oral to completion)
★ Covered (Covered blowjob Without a Condom)Amritsar Call Girls❤️Amritsar Call Girls Service☎️98151-129OO☎️ Call Girl service in Amritsar☎️ ...

❤️Amritsar Call Girls Service☎️98151-129OO☎️ Call Girl service in Amritsar☎️ ...shallyentertainment1
❤️Chandigarh Escort Service☎️9815457724☎️ Call Girl service in Chandigarh☎️ Chandigarh Call Girls Service ☎️ Call Girls In Chandigarh BEST Call Girls in CHANDIGARH Escort Service provide Cute Nice sweet and Sexy Models in beautiful CHANDIGARH city cash in hand to hand call girl in CHANDIGARH and CHANDIGARH escorts. HOT & SEXY MODELS // COLLEGE GIRLS IN CHANDIGARH AVAILABLE FOR COMPLETE ENJOYMENT WITH HIGH PROFILE INDIAN MODEL AVAILABLE HOTEL & HOME ★ SAFE AND SECURE HIGH CLASS SERVICE AFFORDABLE RATE ★ 100% SATISFACTION,UNLIMITED ENJOYMENT. ★ All Meetings are confidential and no information is provided to any one at any cost.
★ EXCLUSIVE Profiles Are Safe and Consensual with Most Limits Respected
★ Service Available In: - HOME & 24x7 :: 3 * 5 *7 *Star Hotel Service .In Call & Out call
Services :
★ A-Level (5 star escort)
★ Strip-tease
★ BBBJ (Bareback Blowjob)Receive advanced sexual techniques in different mode make their life more pleasurable.
★ Spending time in hotel rooms
★ BJ (Blowjob Without a Condom)
★ Completion (Oral to completion)
★ Covered (Covered blowjob Without a Condom)Chandigarh Call Girls❤️Chandigarh Escort Service☎️9815457724☎️ Call Girl service in Chandigarh☎️ C...

❤️Chandigarh Escort Service☎️9815457724☎️ Call Girl service in Chandigarh☎️ C...Rashmi Entertainment
Recently uploaded (20)
💞 Safe And Secure Call Girls Prayagraj 🧿 9332606886 🧿 High Class Call Girl Se...

💞 Safe And Secure Call Girls Prayagraj 🧿 9332606886 🧿 High Class Call Girl Se...
❤️Chandigarh Escorts Service☎️9815457724☎️ Call Girl service in Chandigarh☎️ ...

❤️Chandigarh Escorts Service☎️9815457724☎️ Call Girl service in Chandigarh☎️ ...
Top 20 Famous Indian Female Pornstars Name List 2024

Top 20 Famous Indian Female Pornstars Name List 2024
💞 Safe And Secure Call Girls Coimbatore 🧿 9332606886 🧿 High Class Call Girl S...

💞 Safe And Secure Call Girls Coimbatore 🧿 9332606886 🧿 High Class Call Girl S...
Low Rate Call Girls Pune {9142599079} ❤️VVIP NISHA Call Girls in Pune Maharas...

Low Rate Call Girls Pune {9142599079} ❤️VVIP NISHA Call Girls in Pune Maharas...
Call Girl Service In Mumbai ❤️🍑 9xx000xx09 👄🫦Independent Escort Service Mumba...

Call Girl Service In Mumbai ❤️🍑 9xx000xx09 👄🫦Independent Escort Service Mumba...
💸Cash Payment No Advance Call Girls Hyderabad 🧿 9332606886 🧿 High Class Call ...

💸Cash Payment No Advance Call Girls Hyderabad 🧿 9332606886 🧿 High Class Call ...
🍑👄Ludhiana Escorts Service☎️98157-77685🍑👄 Call Girl service in Ludhiana☎️Ludh...

🍑👄Ludhiana Escorts Service☎️98157-77685🍑👄 Call Girl service in Ludhiana☎️Ludh...
Ulhasnagar Call girl escort *88638//40496* Call me monika call girls 24*

Ulhasnagar Call girl escort *88638//40496* Call me monika call girls 24*
Call Girl In Indore 📞9235973566📞Just Call Inaaya📲 Call Girls Service In Indor...

Call Girl In Indore 📞9235973566📞Just Call Inaaya📲 Call Girls Service In Indor...
❤️Chandigarh Escorts☎️9814379184☎️ Call Girl service in Chandigarh☎️ Chandiga...

❤️Chandigarh Escorts☎️9814379184☎️ Call Girl service in Chandigarh☎️ Chandiga...
Low Rate Call Girls Nagpur {9xx000xx09} ❤️VVIP NISHA Call Girls in Nagpur Mah...

Low Rate Call Girls Nagpur {9xx000xx09} ❤️VVIP NISHA Call Girls in Nagpur Mah...
❤️Chandigarh Escort Service☎️9814379184☎️ Call Girl service in Chandigarh☎️ C...

❤️Chandigarh Escort Service☎️9814379184☎️ Call Girl service in Chandigarh☎️ C...
Gorgeous Call Girls In Pune {9xx000xx09} ❤️VVIP ANKITA Call Girl in Pune Maha...

Gorgeous Call Girls In Pune {9xx000xx09} ❤️VVIP ANKITA Call Girl in Pune Maha...
💚Chandigarh Call Girls Service 💯Jiya 📲🔝8868886958🔝Call Girls In Chandigarh No...

💚Chandigarh Call Girls Service 💯Jiya 📲🔝8868886958🔝Call Girls In Chandigarh No...
❤️Amritsar Call Girls Service☎️98151-129OO☎️ Call Girl service in Amritsar☎️ ...

❤️Amritsar Call Girls Service☎️98151-129OO☎️ Call Girl service in Amritsar☎️ ...
❤️Zirakpur Escorts☎️7837612180☎️ Call Girl service in Zirakpur☎️ Zirakpur Cal...

❤️Zirakpur Escorts☎️7837612180☎️ Call Girl service in Zirakpur☎️ Zirakpur Cal...
❤️Chandigarh Escort Service☎️9815457724☎️ Call Girl service in Chandigarh☎️ C...

❤️Chandigarh Escort Service☎️9815457724☎️ Call Girl service in Chandigarh☎️ C...
❤️ Zirakpur Call Girl Service ☎️9878799926☎️ Call Girl service in Zirakpur ☎...

❤️ Zirakpur Call Girl Service ☎️9878799926☎️ Call Girl service in Zirakpur ☎...
❤️Amritsar Escort Service☎️9815674956☎️ Call Girl service in Amritsar☎️ Amrit...

❤️Amritsar Escort Service☎️9815674956☎️ Call Girl service in Amritsar☎️ Amrit...
Exploring Intensified Seed Train Through Advancements in Perfusion Processing Technologies
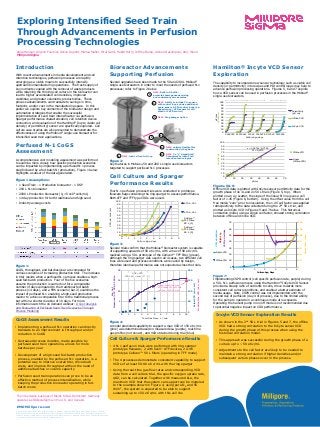
- 1. EMDMillipore.com The life science business of Merck KGaA, Darmstadt, Germany operates as MilliporeSigma in the U.S. and Canada. 0 10 20 30 40 50 60 70 80 90 100 0 1 2 3 4 5 6 7 8 9 VCD,e6cells/mL Culture Age, days ATF-6, 20 L ATF-6, 40 L Cellicon TFF, 50 L (1) Cellicon TFF, 50 L (2) Cellicon™ TFF Filter, 50 L (1) Cellicon™ TFF Filter, 50 L (2) 0 50 100 150 200 0 5 10 15 20 0 1 2 3 4 5 6 7 8 9 10 kLa,hr-1 Measuredin1XPBS,55W/m3 Total Gas Flow, L / min ActualVCD(e6c/mL) ModeledMaxVCD(e6c/mL) model-predicted max VCD capability with this cell line ~150 e6 c/mL OUR = OTR VCD x qO2 = kLa x (C* - CL) Introduction With recent advancements in media development and cell retention technologies, perfusion processes are rapidly emerging as a viable means to successfully intensify upstream biomanufacturing operations. The fresh supply of key nutrients coupled with the removal of waste products while retaining the thriving cell culture in the bioreactor can lead to higher accumulated cell densities, improved viabilities, and greater volumetric productivities. These process advancements can translate to savings in time, footprint, and/or cost in the manufacturing space. In this poster we explore key elements of the bioreactor design and automation strategies that enable the successful implementation of seed train intensification via perfusion. Sparger performance characterization, cell retention device connection, and evaluation of the Hamilton® Incyte viable cell density (or permittivity) sensor are specifically explored. Cell culture case studies are also presented to demonstrate the effectiveness of using the Mobius® single-use bioreactor for intensified seed train applications. Perfused N-1 CoGS Assessment A comprehensive cost modeling assessment was performed to examine more closely how specific production scenarios can be impacted by implementing a perfused N-1 process step followed by a fed-batch N (production). Figure 1 below highlights a subset of the data generated. Figure 1 Assumptions: • 1 Seed Train - 1 Production bioreactor - 1 DSP • 200 L N-1 bioreactor • 2000 L Production bioreactor (X0=5 x106 cells/mL) • 14 day production for both traditional and high seed • Industry average costs Figure 1: CoGS, throughput, and batches/year are compared for various scenarios of increasing production titer. The increase in titer results when a perfused N-1 process enables a high- seed fed-batch production. Four of the five scenarios assume the production is carried out for a comparable number of days compared to the traditional fed-batch process (14 days), and a fifth scenario (bar 2) examines the impact of perfused N-1 enabling a high-seed production reactor to achieve comparable titer to the traditional process but within a shorter duration of 10 days. For more information watch the on demand webinar: A Cost Analysis and Evaluation of Perfused Seed Train Scenarios Through Process Modeling Anne Hansen, Allyson Fournier, Alison Dupont, Marisa Maher, Hiral Gami, Habib Horry, Jeffrey Barna, Arshan Nazempour, Amy Wood MilliporeSigma Hamilton® Incyte VCD Sensor Exploration The capability to incorporate new sensor technology such as viable cell density (or permittivity) into recipes and PID control loops may help to advance perfusion processing operations. Figures 5, 6 and 7 explore how a VCD sensor can be used in perfusion processes in the Mobius® single-use bioreactors. Figures 5 & 6: Offline VCD data is plotted with InCyte sensor permittivity data for the growth phase of 4x 3L and 2x 50 L trials (Figure 5, top). When plotted on an x-y scatter, the slope of the linear fit line gives a cell factor of 1.45 (Figure 6, bottom). Using the offset value from the cell free media “zero” prior to inoculation, the 1.45 cell factor was applied retrospectively to the data collected during the 2nd 50 L trial, and plotted as InCyte VCD in Figures 5 and 7 below. This historical correlation model, using a single cell factor, showed strong correlation between offline and inline VCD. Figure 7: Implementing CSPR control (cell specific perfusion rate, purple) during a 50 L N-1 perfusion process using the Hamilton® InCyte VCD Sensor provided a steady ratio of nutrients to cells, drove towards more consistent cell culture conditions, and enabled a better estimate of media usage. Note, CSPR control was not implemented until the second day of perfusion (day 4 of process), due to the limited ability for the pump to operate in a continuous mode at low speeds. Operating the harvest pump in on/off mode is not recommended due to potential negative impact on CRD performance. Cell Culture & Sparger Performance Results • 4 N-1 perfusion trials were performed with ring sparger prototype flexware; 2 with Xcell™ ATF devices, 2 with prototype Cellicon™ 50 L filters (operating in TFF mode) • The 4 processes demonstrate consistent capability to support VCDs of at least 50-90 e6 c/mL with the ring sparger • Using the real-time gas flow rates and corresponding VCD data from a cell culture trial, the specific oxygen uptake rate, qO2, can be calculated. Together with measured kLa, the maximum VCD that the system can support can be modeled. In the example shown in Figure 4, using pure O2 and 55 W/m3, the system is expected to be able to support sustaining up to 150 e6 c/mL with this cell line Exploring Intensified Seed Train Through Advancements in Perfusion Processing Technologies © 2020 Merck KGaA, Darmstadt, Germany and/or its affiliates. All Rights Reserved. MilliporeSigma, Millipore, Cellicon, Mobius and the vibrant M are trademarks, and the vibrant M are trademarks of Merck KGaA, Darmstadt, Germany or its affiliates. All other trademarks are the property of their respective owners. Detailed information on trademarks is available via publicly accessible resources. Lit. No. MS_PS6000EN V.1.0 Incyte VCD Sensor Exploration Results • As shown in the 2nd 50 L trial in Figures 5 and 7, the offline VCD had a strong correlation to the InCyte sensor VCD during the growth phase of this process when using the historical correlation model. • This approach was successful during the growth phase of a culture up to ~ 50 e6 c/mL • Adjustments to the cell factor are likely to be needed to maintain a strong correlation if higher densities and/or subsequent culture phases occur in the process. 0 20 40 60 80 100 120 140 0 10 20 30 40 50 60 70 0 1 2 3 4 5 6 HarvestRate,L/day VCD,e6cells/mL Weight,kg CSPR,pL/cell.day Culture Age, Days InCyte VCD Offline VCD y = 1,4479x - 0,7763 R² = 0,9773 0 10 20 30 40 50 60 70 80 90 100 0 10 20 30 40 50 60 70 e6cells/mL pF/cm Offline VCD Linear (Offline VCD) 0 10 20 30 40 50 60 70 80 90 100 3456789 01234567 12345 2345678 012345 0123456 pF/cm e6cells/mL Culture Age, days Permittivity (pF/cm) Offline VCD InCyte VCD 50L Trials3L Trials 4 5 NEW: entirely flexible film construction @ bag base NEW: 2 x ¾” AseptiQuik® Connectors to install cell retention technologies 2 NEW: Ability to include CO2 sensor with control loop and use additional spare sensor in PID control loops (ex: Capacitance / VCD sensor for cell bleed or CSPR control) 1 NEW: Built-in X-baffle; promotes homogeneous mixing and consistency across scales 5 4 6 NEW: load cell control loop 3 NEW: Ring sparger option • Implementing a perfused N-1 operation can directly translate to an improvement in throughput and/or reduction in CoGS • Decreased process duration, made possible by perfused seed train operations, allows for more batches per year • Development of a high-seed fed-batch production process, enabled by the perfused N-1 operation, is a potential way to improve overall titer, drive down costs, and improve throughput without the need of additional batches or volume capacity • Perfusion seed train operations can prove to be an effective method of process intensification, while keeping the production bioreactor operating in fed- batch mode CoGS Assessment Results Bioreactor Advancements Supporting Perfusion Several upgrades have been made to the 50 and 200 L Mobius® single-use bioreactors in order to meet the needs of perfused N-1 processes; refer to Figure 2 below. Figure 2: Key features in Mobius® 50 and 200 L single-use bioreactors targeted to support perfused N-1 processes Cell Culture and Sparger Performance Results Four N-1 perfusion process trials were conducted in prototype flexware bags containing the ring sparger to assess performance. Both ATF and TFF type CRDs were used. Figure 3: Several trials confirm that the Mobius® bioreactor system is capable of supporting upwards of 50 e6 c/mL, with a max of 90 e6 c/mL realized using a 50 L prototype of the Cellicon™ TFF filter (green). Although the ring sparger was used in all 4 cases, two different cell lines were used and process conditions were varied by design, therefore identical performance was not expected across the trials. Figure 4: A model predicted capability to support a max VCD of 150 e6 c/mL (pink) was determined based on measured kLa (purple), real-time gas profiles (not shown), and VCD obtained during trial (blue)
