How does ZMapp function to fight Ebola in humans & can it be used as a large-scale treatment method?
•Download as PPTX, PDF•
2 likes•1,527 views
A brief overview of ZMapp & its potential in large-scale treatment for Ebola in humans, by Michael Small, Ping Chi Liu, Zehraa Cheaib, Sheldon Pereira, & Raj Shah at McMaster University, Immunology, Fall of 2014.
Report
Share
Report
Share
Recommended
More Related Content
Recently uploaded
Recently uploaded (20)
call girls in Connaught Place DELHI 🔝 >༒9540349809 🔝 genuine Escort Service ...

call girls in Connaught Place DELHI 🔝 >༒9540349809 🔝 genuine Escort Service ...
Russian Call Girl Brookfield - 7001305949 Escorts Service 50% Off with Cash O...

Russian Call Girl Brookfield - 7001305949 Escorts Service 50% Off with Cash O...
Call Girls Frazer Town Just Call 7001305949 Top Class Call Girl Service Avail...

Call Girls Frazer Town Just Call 7001305949 Top Class Call Girl Service Avail...
Call Girls Jayanagar Just Call 7001305949 Top Class Call Girl Service Available

Call Girls Jayanagar Just Call 7001305949 Top Class Call Girl Service Available
Call Girls Kanakapura Road Just Call 7001305949 Top Class Call Girl Service A...

Call Girls Kanakapura Road Just Call 7001305949 Top Class Call Girl Service A...
Call Girls Service In Shyam Nagar Whatsapp 8445551418 Independent Escort Service

Call Girls Service In Shyam Nagar Whatsapp 8445551418 Independent Escort Service
Hemostasis Physiology and Clinical correlations by Dr Faiza.pdf

Hemostasis Physiology and Clinical correlations by Dr Faiza.pdf
Housewife Call Girls Bangalore - Call 7001305949 Rs-3500 with A/C Room Cash o...

Housewife Call Girls Bangalore - Call 7001305949 Rs-3500 with A/C Room Cash o...
Book Call Girls in Kasavanahalli - 7001305949 with real photos and phone numbers

Book Call Girls in Kasavanahalli - 7001305949 with real photos and phone numbers
Call Girls Service Noida Maya 9711199012 Independent Escort Service Noida

Call Girls Service Noida Maya 9711199012 Independent Escort Service Noida
Bangalore Call Girls Marathahalli 📞 9907093804 High Profile Service 100% Safe

Bangalore Call Girls Marathahalli 📞 9907093804 High Profile Service 100% Safe
Call Girl Koramangala | 7001305949 At Low Cost Cash Payment Booking

Call Girl Koramangala | 7001305949 At Low Cost Cash Payment Booking
High Profile Call Girls Jaipur Vani 8445551418 Independent Escort Service Jaipur

High Profile Call Girls Jaipur Vani 8445551418 Independent Escort Service Jaipur
Call Girls Hebbal Just Call 7001305949 Top Class Call Girl Service Available

Call Girls Hebbal Just Call 7001305949 Top Class Call Girl Service Available
College Call Girls Pune Mira 9907093804 Short 1500 Night 6000 Best call girls...

College Call Girls Pune Mira 9907093804 Short 1500 Night 6000 Best call girls...
Call Girls Thane Just Call 9910780858 Get High Class Call Girls Service

Call Girls Thane Just Call 9910780858 Get High Class Call Girls Service
call girls in green park DELHI 🔝 >༒9540349809 🔝 genuine Escort Service 🔝✔️✔️

call girls in green park DELHI 🔝 >༒9540349809 🔝 genuine Escort Service 🔝✔️✔️
Call Girls Service Chennai Jiya 7001305949 Independent Escort Service Chennai

Call Girls Service Chennai Jiya 7001305949 Independent Escort Service Chennai
Call Girls Jp Nagar Just Call 7001305949 Top Class Call Girl Service Available

Call Girls Jp Nagar Just Call 7001305949 Top Class Call Girl Service Available
Dwarka Sector 6 Call Girls ( 9873940964 ) Book Hot And Sexy Girls In A Few Cl...

Dwarka Sector 6 Call Girls ( 9873940964 ) Book Hot And Sexy Girls In A Few Cl...
Featured
More than Just Lines on a Map: Best Practices for U.S Bike Routes

More than Just Lines on a Map: Best Practices for U.S Bike RoutesProject for Public Spaces & National Center for Biking and Walking
Featured (20)
Content Methodology: A Best Practices Report (Webinar)

Content Methodology: A Best Practices Report (Webinar)
How to Prepare For a Successful Job Search for 2024

How to Prepare For a Successful Job Search for 2024
Social Media Marketing Trends 2024 // The Global Indie Insights

Social Media Marketing Trends 2024 // The Global Indie Insights
Trends In Paid Search: Navigating The Digital Landscape In 2024

Trends In Paid Search: Navigating The Digital Landscape In 2024
5 Public speaking tips from TED - Visualized summary

5 Public speaking tips from TED - Visualized summary
Google's Just Not That Into You: Understanding Core Updates & Search Intent

Google's Just Not That Into You: Understanding Core Updates & Search Intent
The six step guide to practical project management

The six step guide to practical project management
Beginners Guide to TikTok for Search - Rachel Pearson - We are Tilt __ Bright...

Beginners Guide to TikTok for Search - Rachel Pearson - We are Tilt __ Bright...
Unlocking the Power of ChatGPT and AI in Testing - A Real-World Look, present...

Unlocking the Power of ChatGPT and AI in Testing - A Real-World Look, present...
More than Just Lines on a Map: Best Practices for U.S Bike Routes

More than Just Lines on a Map: Best Practices for U.S Bike Routes
Ride the Storm: Navigating Through Unstable Periods / Katerina Rudko (Belka G...

Ride the Storm: Navigating Through Unstable Periods / Katerina Rudko (Belka G...
How does ZMapp function to fight Ebola in humans & can it be used as a large-scale treatment method?
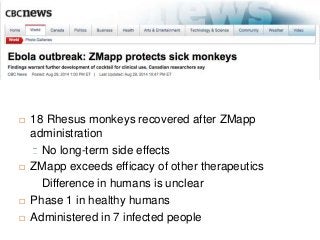
- 1. 18 Rhesus monkeys recovered after ZMapp administration No long-term side effects ZMapp exceeds efficacy of other therapeutics Difference in humans is unclear Phase 1 in healthy humans Administered in 7 infected people
- 2. Research Question How does ZMapp function to fight Ebola in humans and can it be used as a large-scale treatment method? Introduction to ZMapp • Produced in the leaves of Nicotiana plants • 3 different humanized MAbs → c13C6, c2G4 & c4G7
- 3. Mechanism of ZMapp MAbs bind to EBV epitope via glycoproteins EBV-bound MAbs lead to c1q and FcγRI interaction C1q activation leads to complement response FcγRI activation lead to adaptive immune response, such as CD8+ T-cell activation
- 4. Analysis of Implications Time of administration As late as 5 days after exposure Can detect Ebola virus at this time Limited testing capacity in Africa Feasibility 20-40 ZMapp doses per month Plans to ramp up production in the works
- 5. Analysis of Implications Efficacy 5 out of 7 Ebola patients recovered after administration of ZMapp 45% of patients recovered without treatment Glycoprotein mutation in the Ebola virus Ethical concerns Conclusion The article is justified as ZMapp has potential of impeding progress of Ebola virus but must be used with precaution until further clinical trials and safety studies are conducted.
