Protein metabolism, july'20
•
9 likes•1,133 views
Protein turnover, Amino acid pool, Protein metabolism
Report
Share
Report
Share
Recommended
Recommended
More Related Content
What's hot
What's hot (20)
Metabolism of Basic Amino Acids (Arginine, Histidine, Lysine)

Metabolism of Basic Amino Acids (Arginine, Histidine, Lysine)
Amino acid metabolism | Transamination | Deamination |

Amino acid metabolism | Transamination | Deamination |
Nitrogen metabolism (metabolic fate of amino acid, catabolism of amino acid, ...

Nitrogen metabolism (metabolic fate of amino acid, catabolism of amino acid, ...
Digestion protein, absorption amino acid and amino acid pool 

Digestion protein, absorption amino acid and amino acid pool
Similar to Protein metabolism, july'20
Similar to Protein metabolism, july'20 (20)
Formation of ammonia and Urea cycle by Dr. Ashok Kumar J

Formation of ammonia and Urea cycle by Dr. Ashok Kumar J
Catabolism of Amino Acids.ppt degradation of aminoacids

Catabolism of Amino Acids.ppt degradation of aminoacids
More from enamifat
More from enamifat (20)
Central Dogma, Gene, Genetic Code, Codon, Genome, Genotype, Phenotype , Trai...

Central Dogma, Gene, Genetic Code, Codon, Genome, Genotype, Phenotype , Trai...
Structure of dna, its organization & functions, july 2020

Structure of dna, its organization & functions, july 2020
Introduction to nucleic acid, chemistry of nucleotiides , july 2020

Introduction to nucleic acid, chemistry of nucleotiides , july 2020
Recently uploaded
Recently uploaded (20)
Call Girls Coimbatore Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Coimbatore Just Call 8250077686 Top Class Call Girl Service Available
VIP Hyderabad Call Girls Bahadurpally 7877925207 ₹5000 To 25K With AC Room 💚😋

VIP Hyderabad Call Girls Bahadurpally 7877925207 ₹5000 To 25K With AC Room 💚😋
Premium Call Girls In Jaipur {8445551418} ❤️VVIP SEEMA Call Girl in Jaipur Ra...

Premium Call Girls In Jaipur {8445551418} ❤️VVIP SEEMA Call Girl in Jaipur Ra...
Russian Call Girls Service Jaipur {8445551418} ❤️PALLAVI VIP Jaipur Call Gir...

Russian Call Girls Service Jaipur {8445551418} ❤️PALLAVI VIP Jaipur Call Gir...
Call Girls Service Jaipur {8445551418} ❤️VVIP BHAWNA Call Girl in Jaipur Raja...

Call Girls Service Jaipur {8445551418} ❤️VVIP BHAWNA Call Girl in Jaipur Raja...
Call Girls Rishikesh Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Rishikesh Just Call 8250077686 Top Class Call Girl Service Available
Call Girls in Delhi Triveni Complex Escort Service(🔝))/WhatsApp 97111⇛47426

Call Girls in Delhi Triveni Complex Escort Service(🔝))/WhatsApp 97111⇛47426
Top Rated Pune Call Girls (DIPAL) ⟟ 8250077686 ⟟ Call Me For Genuine Sex Serv...

Top Rated Pune Call Girls (DIPAL) ⟟ 8250077686 ⟟ Call Me For Genuine Sex Serv...
(Low Rate RASHMI ) Rate Of Call Girls Jaipur ❣ 8445551418 ❣ Elite Models & Ce...

(Low Rate RASHMI ) Rate Of Call Girls Jaipur ❣ 8445551418 ❣ Elite Models & Ce...
Top Rated Call Girls Kerala ☎ 8250092165👄 Delivery in 20 Mins Near Me

Top Rated Call Girls Kerala ☎ 8250092165👄 Delivery in 20 Mins Near Me
Independent Call Girls In Jaipur { 8445551418 } ✔ ANIKA MEHTA ✔ Get High Prof...

Independent Call Girls In Jaipur { 8445551418 } ✔ ANIKA MEHTA ✔ Get High Prof...
Call Girls Mumbai Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Mumbai Just Call 8250077686 Top Class Call Girl Service Available
Call Girls Hyderabad Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Hyderabad Just Call 8250077686 Top Class Call Girl Service Available
Jogeshwari ! Call Girls Service Mumbai - 450+ Call Girl Cash Payment 90042684...

Jogeshwari ! Call Girls Service Mumbai - 450+ Call Girl Cash Payment 90042684...
Top Rated Hyderabad Call Girls Chintal ⟟ 9332606886 ⟟ Call Me For Genuine Se...

Top Rated Hyderabad Call Girls Chintal ⟟ 9332606886 ⟟ Call Me For Genuine Se...
Call Girls Madurai Just Call 9630942363 Top Class Call Girl Service Available

Call Girls Madurai Just Call 9630942363 Top Class Call Girl Service Available
Top Quality Call Girl Service Kalyanpur 6378878445 Available Call Girls Any Time

Top Quality Call Girl Service Kalyanpur 6378878445 Available Call Girls Any Time
Andheri East ) Call Girls in Mumbai Phone No 9004268417 Elite Escort Service ...

Andheri East ) Call Girls in Mumbai Phone No 9004268417 Elite Escort Service ...
Call Girl In Pune 👉 Just CALL ME: 9352988975 💋 Call Out Call Both With High p...

Call Girl In Pune 👉 Just CALL ME: 9352988975 💋 Call Out Call Both With High p...
Protein metabolism, july'20
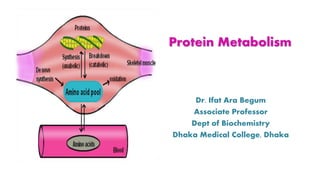
- 1. Protein Metabolism Dr. Ifat Ara Begum Associate Professor Dept of Biochemistry Dhaka Medical College, Dhaka
- 2. 2
- 3. 3
- 4. 4
- 5. 5
- 7. 7 Protein turnover is the rate at which body proteins are constantly being degraded and again resynthesized. It is 150-300 gm/day in adult i.e. 1-2% of total body protein in adult. [Total body protein in 70kg adult male is 12-14 kg]
- 8. 8 Catabolized portion (20%) must be replaced by dietary protein to maintain normal body protein content & nitrogen balance
- 10. 10
- 11. 11 Nitrogen balance is the difference between nitrogen intake & nitrogen loss in an individual. Three types: i) Nitrogen Equilibrium: In normal adults ii) Positive Nitrogen Balance : During growth, pregnancy iii) Negative Nitrogen Balance : DM, malignancy, TB, Trauma, Surgery etc If loss exceeds 30% of total protein: Fatal
- 12. Amino Acid Pool
- 13. What is Amino Acid Pool? 13 It is the free amino acid content distributed throughout the extracellular fluid Quantitatively, it is about 100 gm in an adult individual. 50% : Glutamate & Glutamine 10% : Essential Amino Acids 40% : Other Amino Acids
- 14. 14
- 16. What is Amino Acid? 16 Amino group containing carboxylic acid
- 17. 17
- 18. 18 Anabolism Catabolism Synthesis of: NEAA Protein NPN subs Urea from NH3 via urea cycle Glucose, glycogen FA, fat, steroid, ketone bodies Transamination Deamination & removal of NH2 group as NH3 Catabolism of amino acid carbon - skeleton to : - Pyruvate - Acetyl CoA - Intermediates of TCA cycle Oxidation of Catabolic end products of amino acid carbon skeleton via TCA cycle
- 19. Transamination
- 20. Site of transamination : Cytoplasm of: Liver Kidney Heart Sk. Muscle Brain. Rate limiting enzyme: Aminotransferase Coenzyme needed: Pyridoxal phosphate which acts as an intermediate carrier of an NH2 group. Nature: Amphibolic & reversible Substrate: One amino acid & one keto acid Product: One keto acid & one amino acid .
- 21. What is Transamination? 21 It is the transfer of NH2 group from an amino acid to a keto acid with simultaneous production of corresponding keto acid & amino acid respectively. Remember, No free NH3 is produced here Only transfer of NH2 group occurs
- 22. Amino acid participating in transamination : All Amino acids except: Lys (Lysine) Thr (Threonine) Pro (Proline) Keto acid participating in transamination: Three keto acids mostly participates: α- ketoglutarate (ketoacid of glutamate) Oxaloacetate (keto acid of aspartate) Pyruvate (ketoacid of alanine)
- 23. 23
- 24. Example of Aminotransferase / Transaminase Alanine transaminase Aspartate transaminase Glutamate transaminase. Clinical importance of these enzymes: Role as hepatic marker / cardiac marker
- 25. 25
- 26. Provides a link b/w carbohydrate, protein & fat metabolism Biosynthesis of NEAA Importance of Transamination Funneling of NH2 group of different amino acids ultimately to α-KG. α-KG will form Glutamate (The major amino acid that undergoes oxidative deamination) Formation of C skeleton / keto acid of an amino acid: which can be catabolized & oxidized to generate energy
- 28. Site of oxidative deamination: Mitochondria of: Liver Kidney Heart Sk. Muscle Rate limiting enzyme: Glutamate dehydrogenase Coenzyme needed: NAD. Nature: Catabolic Substrate: Glutamate Product: NH3 & α-KG Types: Oxidative & Nonoxidative .
- 29. What is Oxidative Deamination? 29 Removal of –NH2 group from an amino acid in the form of free NH3 with simultaneous formation of its corresponding keto acid (C-skeleton) Amino acid unique in the process of deamination: Glutamate Because, it is the only amino acid that undergoes rapid oxidative deamination catalyzed by glutamate dehydrogenase (an active dehydrogenase)
- 30. 30
- 31. 31
- 32. Channel nitrogen of AA as ammonia to urea cycle Generate C- skeleton of AA for catabolic purpose Importance of Deamination
- 34. 34
- 35. Sources of Ammonia Bacterial degradation of urea into NH3 (by bacterial urease in intestinal lumen) Catabolism of amino acids ( via Transamination & deamination) Catabolism of purine & pyrimidine nitrogenous bases Catabolism of Glutamine (& Glutamate)
- 36. 36 Glutamine : The temporary non-toxic storage & transport form of NH3.
- 37. 37
- 38. Disposal / Metabolic fates of Ammonia Excretion of NH3 with urine as NH4+ salt Formation of urea in urea cycle & its excretion in urine Formation of Glutamate & Glutamine in liver, kidney, muscle, brain
- 39. 39
- 40. Toxicity which results from hyperammonemia (Normal level : 5- 50μmol/L Brain tissue is mostly affected & there is depression in cerebral activity Ammonia Intoxication Causes of hyperammonemia: Hepatic dysfunction (Cirrhosis of liver, hepatic failure) Deficiency of enzyme of the urea cycle
- 41. Biochemical basis of ammonia intoxication / Hepatic encephalopathy 41 When excessive amounts of ammonia enter the central nervous system, the brain's defences are severely challenged
- 42. 42
- 43. 43
- 45. Urea cycle
- 46. Site of Urea cycle : Mitochondria & cytoplasm of liver Rate limiting enzyme: Carbamoylphosphate synthase I Nature: Anabolic Substrate: NH3 (Two NH3 participates , one directly and another from aspartate) Product: Urea (NH2 – CO – NH2) .
- 47. 47
- 48. 48
- 49. 49
- 50. Interaction with TCA cycle Disposal of NH3 & CO2 Functions / Importance of urea cycle Conversion of toxic ammonia into nontoxic urea Synthesis of NEAA: Arginine Proline From ornithine
- 51. 51
- 52. 52
- 53. Inborn error of protein metabolism 53 Alkaptonuria Homocystinuria Phenylketonuria Albinism Maple syrup urine disease
- 54. 54