Neuromuscular blockade & Reversal agents & Monitoring
- 1. Resident: B.Ankhzaya (MNUMS) Neuromuscular Blocking Agents
- 2. 1. Physiological mechanism of NMBD 2. Classification 3. Depolarizing blocking drugs 4. Nondepolarizing drugs 5. Drug Interaction 6. Altered responses to NMBD 7. Reversal of Neuromuscular blockade 8. Monitoring Neuromuscular blockade Content
- 3. PHYSIOLOGICAL MECHANISM OF NMBD
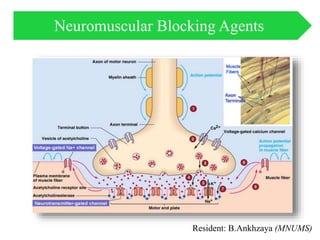
- 4. Nerve Stimulation. Under resting conditions, the electrical potential of the inside of a nerve cell is negative with respect to the outside (typically –90 mV). Release of ACh into the synaptic cleft occurs when an action potential arrives at the nerve terminal (neuromuscular junction [NMJ]
- 6. CLASSIFICATION NMBD Depolarizing agents Nondepolarizing agents NMBDs interact with the ACh receptor either by depolarizing the end plate by competing with ACh for binding sites. • Succinylcholine • Atracurium • Cisatracurium • Rocuronium • Vecuronium • Doxacurium • Pancuronium
- 7. DEFINITION OF NEUROMUSCULAR BLOCKING DRUGS ACCORDING TO THE ONSET AND DURATION OF BLOCK AT THE ADDUCTOR POLLICIS Onset to Maximum Block Ultra rapid ( <1min) Succinylcholine Rapid (1–2 min) Rocuronium Intermediate (2–4 min) Atracurium Vecuronium Pancuronium Long (>4 min) Cisatracurium Doxacurium Tubocurarine Duration to 25% Recovery of T1 Ultra short (<8 min) Succinylcholine Intermediate (20–50 min) Atracurium Cisatracurium Rocuronium Vecuronium Long (>50 min) Doxacurium Pancuronium Tubocurarine
- 8. DEPOLARIZING BLOCKING DRUGS (SUCCINYLCHOLINE) • 20mg/ml, 100mg/ml- 10,20 ml • Succinylcholine (SCh) remains a useful muscle relaxant because of its ultra-rapid onset and short- duration • Neuromuscular Effects-The net effect of SCh -induced depolarization is uncoordinated skeletal muscle activity that manifests clinically as fasciculations • R-Postsynaptic nicotinic receptors, Extrajunctional receptors and Presynaptic receptors
- 9. • SCh predictably increases masseter muscle tone (this may be responsible for poor intubating conditions), and masseter muscle spasm may be associated with malignant hyperthermia. Pharmacokinetic SCh is rapidly hydrolyzed (the elimination half-life of <1 min) by plasma cholinesterase (pseudocholinesterase) within the synaptic cleft The dose producing 95% blockade (ED95) at the adductor pollicis is 0.3 to 0.5 mg/kg. The time until full recovery at the adductor pollicis muscle is dose dependent and reaches 10 to 12 minutes after a dose of 1 mg/kg
- 10. Dose Tracheal intubation 1 mg/kg (the usual dose) +нэмэлт тун анхны тунгын 25% Нийт тунг 4-6 мг/кг аас хэтрүүлэхгүй intravenous (IV) which is increased to 1.5– 2.0 mg/kg IV if pretreatment is used). 4 mg/kg (2-3mg/kg) Intramuscular(IM) SCh is the only effective NMBD in children with difficult intravenous access and provides adequate intubating conditions in about 4 minutes. Onset time 0.5min Duration of action 5-10min SCh is especially indicated for “rapid sequence induction” when a patient presents with a full stomach and the possibility of aspiration of gastric contents (fast onset and brief duration allowing return of spontaneous breathing).
- 12. Side effect of SCh
- 13. Tell your doctor or get medical help right away if you have any of the following signs or symptoms that may be related to a very bad side effect: • Signs of an allergic reaction, like rash; hives; itching; red, swollen, blistered, or peeling skin with or without fever; wheezing; tightness in the chest or throat; trouble breathing or talking; unusual hoarseness; or swelling of the mouth, face, lips, tongue, or throat. • Signs of a high potassium level like a heartbeat that does not feel normal; change in thinking clearly and with logic; feeling weak, lightheaded, or dizzy; feel like passing out; numbness or tingling; or shortness of breath. • Slow heartbeat. • Very bad dizziness or passing out. • Very bad headache. • Muscle pain. • Twitching. • Not able to pass urine or change in how much urine is passed. • A heartbeat that does not feel normal. • Chest pain or pressure. • More eye pressure. • Trouble breathing, slow breathing, or shallow breathing. • This medicine may cause a very bad and sometimes deadly problem called malignant hyperthermia. Call your doctor right away if you have a fast heartbeat, fast breathing, fever, or spasm or stiffness of the jaw muscles.
- 14. NONDEPOLARIZING DRUGS Benzylisoquinolinium compound • Atracurium • Mivacurium • Doxacurium • Cisatracurium • Tubocurarine • Alcuronium Aminosteroid compound • Pancuronium • Vecuronium • Pipecuronium • Rocuronium • Rapacuronium
- 15. Pharmacokinetic • The pharmacokinetic variables derived from measurements of plasma concentrations of nondepolarizing muscle relaxants depend on the dose administered, the sampling schedule used, and the accuracy of the assay. • All nondepolarizing muscle relaxants have a volume of distribution that is approximately equal to extracellular fluid volume.
- 17. COMPARATIVE PHARMACOLOGY OF NONDEPOLARIZING MUSCLE RELAXANTS
- 18. • 10mg/ml – 5,10 ml solution • Metabolism is by nonspecific ester hydrolysis (group of tissue esterases that are distinct from plasma or acetyl cholinesterases with the same tissue esterases ) and the Hofmann reaction (nonenzymatic degradation at body temperature and pH) • An end product of degradation of atracurium is laudanosine. (High doses in animals cause seizures, but no deleterious effect has been conclusively documented in humans.) ATRACURIUM
- 19. Монгол номон дээр: Dose Onset Duration Tracheal intubation 0,3-0,6мг/кг 2min 20-40min Add dose 0,1-0,2mg/kg Infusion 0,3-0,6mg/kg/h It has no direct cardiovascular effect but may release histamine
- 20. • 2,10mg/ml- 5,10,20 ml injection • • It is devoid of histamine-releasing properties even at high doses. • About 77% of the drug undergoes Hofmann degradation, and 15% is excreted unchanged in the urine. • Renal failure is associated with a slight reduction in its plasma clearance, but its duration of action is not prolonged. As a lower dose is given, it produces less laudanosine than an equipotent dose of atracurium. CISATRACURIUM
- 21. Dose Onset Duration Tracheal intubation 0,1-0,2mg/kg 2min 30-40min Aad dose 0,03mg/kg Infusion 0,06-0,18mg/kg/h
- 22. • 10mg/ml-5,10 ml injection • An aminosteroid NMBD that has a more rapid onset (intubating conditions 60 seconds after administration of 1 mg/kg IV resemble conditions after administration of 1 mg/kg IV of SCh) but similar duration of action and pharmacokinetic characteristics as vecuronium( but tachycardia). ROCURONIUM
- 23. • Pharmacology. As for other short- and intermediateacting NMBDs, the onset of action of rocuronium is more rapid at the diaphragm and laryngeal muscle than at the adductor pollicis, and about twice as much drug is required to produce the same degree of paralysis. • Allergy. The incidence of anaphylactic reactions to rocuronium is unclear, and current data suggest marked variations in the geographical incidence (higher in France than the United States), perhaps reflecting sensitization to pholcodine, an antitussive in cough syrups. • Rapid Sequence Induction. Rocuronium is the drug of choice for rapid sequence induction (>1 mg/kg resulting in prolonged duration of action) and tracheal intubation if SCh is contraindicated (children with undiagnosed muscle dystrophy).
- 24. Dose Onset Duration Tracheal intubation 0,6mg/kg About 1min 30-40min Aad dose 0,15mg/kg
- 25. • 1mg/ml-10,20 ml injection • Intermediate-acting aminosteroid NMBD that is devoid of histamine- releasing and cardiovascular side effects. (It has been largely replaced by rocuronium.) • NOT effect BP or cardiac output VECURONIUM
- 26. Dose Onset Duration Tracheal intubation 0,08-0,1mg/kg 2-3min 20-30min Add dose 0,03-0,05mg/kg
- 27. • 1mg/ml-10ml • long-acting nondepolarizing NMBD with a steroid structure but lacking any endocrine effects. PANCURONIUM
- 28. • Cardiovascular Effects:Pancuronium is associated with increases in heart rate, blood pressure, and cardiac output, especially at doses >2 × ED95. • Pancuronium does not release histamine. • Clinical Use. The slow onset of action of pancuronium limits its usefulness in facilitating tracheal intubation. • In cardiac anesthesia, this drug has enjoyed popularity because it counters the bradycardic effects of high doses of opioids. • Pancuronium neuromuscular blockade is more difficult to reverse than blockade of the intermediate-acting nondepolarizing NMBDs
- 29. Dose Onset Duration Tracheal intubation 0,1mg/kg 3min 30-60min Add dose 0,01-0,02mg/kg
- 30. DRUG INTERACTION
- 31. ALTERED RESPONSES TO NEUROMUSCULAR BLOCKING AGENTS
- 32. Cholinesterase Inhibitors & Other Pharmacologic Antagonists to Neuromuscular Blocking Agents
- 36. • Cholinesterase inhibitors indirectly increase the amount of acetylcholine available to compete with the nondepolarizing agent, thereby reestablishing normal neuromuscular transmission. Mechanism of Action
- 37. Pharmacokinetic
- 38. CVS The predominant effect on the heart is bradycardia caused by the accumulation of acetylcholine. This can result in a decrease in cardiac output and blood pressure. RS Anticholinesterases cause bronchial smooth muscle contraction leading to bronchospasm and hypoxia, which is aggravated by an increase in secretions GIS Oesophageal motility, gastric motility and production of gastric secretions are enhanced. Also, anticholinesterases augment the motor activity of the small and large bowel. In high doses, they can lead to vomiting, diarrhoea and incontinence. Eye On local application, anticholinesterases cause constriction of the sphincter pupillae and ciliary muscles leading to miosis and blocking of the accommodation reflex. Intraocular pressure, if elevated, usually decreases as a result of facilitation of the outflow of aqueous humour. Secretory glands Anticholinesterases increase the activity of all secretory glands innervated by postganglionic cholinergic fibres, i.e. bronchial, salivary, sweat, lacrimal, gastric, intestinal and pancreatic glands Neuromuscular Somatic nerve (miasteni) Pharmacological properties
- 39. NEOSTIGMINE • 0,25; 0,5; 1mg/ml-5,10ml solution • 0,5%-eye drops • The former provides covalent bonding to acetylcholinesterase • lipid insoluble, so that it cannot pass through the blood–brain barrier.
- 40. Cholinesterase Inhibitor Usual Dose of Cholinesterase Inhibitor Recommended Anticholinergic Usual Dose of Anticholinergic per mg of Cholinesterase Inhibitor (1mg) Neostigmine 0.04–0.08 mg/kg Glycopyrrolate than atropine 0.2 mg Onset time <1min or 5min Peak effect 10min Duration of action 20-30 min or last more 1 hr Neostigmine is metabolized by plasma esterases; 60% of the drug is excreted in urine. In the presence of renal impairment, plasma clearance is reduced and the elimination half-life prolonged.
- 41. • It has been reported that neostigmine crosses the placenta, resulting in fetal bradycardia.Thus, theoretically, atropine may be a better choice of an anticholinergic agent than glycopyrrolate in pregnant patients receiving neostigmine, but there is no evidence that this makes any difference in patient outcomes. • Neostigmine is also used to treat myasthenia gravis, urinary bladder atony, and paralytic ileus. • It has been shown to cause an increase in postoperative nausea and vomiting
- 42. • Solution of 5 mg/ml-2ml • Tab -60mg • Pyridostigmine is 20% as potent as neostigmine • It is similar to neostigmine in that it binds to acetylcholinesterase via a covalent bond and is lipid insoluble. PYRIDOSTIGMINE
- 43. Cholinesterase Inhibitor Usual Dose of Cholinesterase Inhibitor Recommended Anticholinergic Usual Dose of Anticholinergic per mg of Cholinesterase Inhibitor (1mg) Pyridostigmine 0.1–0.25 mg/kg Glycopyrrolate 0.05 mg Atropine 0,1mg Onset time 10-15min Duration of action 6h or >2 hour
- 44. • Solution 10mg/ml • It is available with atropine as a combination (Enlon-Plus; 10 mg edrophonium and 0.14 mg atropine per mL) • Edrophonium is less than 10% as potent as neostigmine. • limits lipid solubility • The drug competes with acetylcholine and binds by a non-covalent bond to acetylcholinesterase at the anionic site EDROPHONIUM
- 45. Cholinesterase Inhibitor Usual Dose of Cholinesterase Inhibitor Recommended Anticholinergic Usual Dose of Anticholinergic per mg of Cholinesterase Inhibitor (1mg) Edrophonium 0.5–1 mg/kg Atropine 0.014 mg Onset time 1-2min Peak effect 0,8-2,0 min Duration of action 10min • Higher doses prolong the duration of action to more than 1 hr. • Although glycopyrrolate (0.007 mg per 1 mg of edrophonium) can also be used, it should be given several minutes prior to edrophonium to avoid the possibility of bradycardia.
- 46. • Solution –1mg/ml • It is lipid soluble and is the only clinically available cholinesterase inhibitor that freely passes the blood– brain barrier PHYSOSTIGMINE
- 47. Cholinesterase Inhibitor Usual Dose of Cholinesterase Inhibitor Recommended Anticholinergic Usual Dose of Anticholinergic per mg of Cholinesterase Inhibitor (1mg) Physostigmine 0.01–0.03 mg/kg Usually not necessary NA Physostigmine is metabolized by plasma esterases; elimination does not depend on renal excretion, unlike the other anticholinesterases.
- 48. • The lipid solubility and central nervous system penetration of physostigmine limit its usefulness as a reversal agent for nondepolarizing blockade, but make it effective in the treatment of central anticholinergic toxicity caused by overdoses of atropine or scopolamine. • In addition, it reverses some of the central nervous system depression and delirium associated with use of benzodiazepines and volatile anesthetics. • Physostigmine (0.04 mg/kg) has been shown to be effective in preventing postoperative shivering. • It reportedly partially antagonizes morphine-induced respiratory depression, presumably because morphine reduces acetylcholine release in the brain. These effects are transient, and repeated doses may be required. • Other possible muscarinic side effects include excessive salivation, vomiting, and convulsions.
- 49. • 100mg/ml -2; 5 ml injection • This cyclodextrin leads to restoration of normal neuromuscular function by selectively binding to rocuronium (and to a lesser extent to vecuronium and pancuronium) SUGAMMADEX
- 50. Cholinesterase Inhibitor Usual Dose of Cholinesterase Inhibitor Sugammadex 4-8 mg/kg • Sugammadex and sugammadex– rocuronium complexes are excreted unchanged via the kidneys. (Clearance of sugammadex decreased markedly in patients with renal failure.) • With an injection of 8 mg/kg, given 3 min after administration of 0.6 mg/kg of rocuronium, recovery of TOF ratio to 0.9 was observed within 2 min. • It produces rapid and effective reversal of both shallow and profound rocuronium-induced neuromuscular blockade.
- 51. • Because of some concerns about hypersensitivity and allergic reactions, sugammadex has not yet been approved by the US Food and Drug Administration • It is devoid of cardiovascular and other side effects. Block- produced by SCh or any of the benzylisoquinolines (atracurium, cisatracurium, mivacurium) is unaffected by sugammadex.
- 52. • Exogenous administration of L -cysteine (10–50 mg/kg intravenously) given to anesthetized monkeys 1 min after these neuromuscular blocking agents abolished the block within 2–3 min. • This unique method of antagonism by adduct formation and inactivation is still in the investigative stage, especially in terms of its safety and efficacy in humans. L -CYSTEINE
- 53. • Anticholinergics are esters of an aromatic acid combined with an organic base • This competitively blocks binding by acetylcholine and prevents receptor activation. Anticholinergic Drugs (Antimuscarinic)
- 54. • Blockade of muscarinic receptors in the sinoatrial node produces tachycardia. THis effect is especially useful in reversing bradycardia due to vagal reflexes (eg, baroreceptor reflex, peritoneal traction, or oculocardiac reflex). • A transient slowing of heart rate in response to smaller intravenous doses of atropine(<0.4 mg) has been reported . • Atrial arrhythmias and nodal (junctional) rhythms occasionally occur . • Anticholinergics generally have little effect on ventricular function or peripheral vasculature because of the paucity of direct cholinergic innervation of these areas despite the presence of cholinergic receptors. • Presynaptic muscarinic receptors on adrenergic nerve terminals are known to inhibit norepinephrine release, so muscarinic antagonists may modestly enhance sympathetic activity. • Large doses of anticholinergic agents can result in dilation of cutaneous blood vessels (atropine flush) CVS
- 55. RS The anticholinergics inhibit the secretions of the respiratory tract mucosa, from the nose to the bronchi, a valuable property during airway endoscopic or surgical procedures. Relaxation of the bronchial smooth musculature reduces airway resistance and increases anatomic dead space. These effects are particularly pronounced in patients with chronic obstructive pulmonary disease or asthma. CNS Cerebral stimulation may present as excitation, restlessness, or hallucinations. Cerebral depression, including sedation and amnesia, are prominent after scopolamine. Physostigmine, a cholinesterase inhibitor that crosses the blood–brain barrier, promptly reverses anticholinergic actions on the brain GIS Salivary secretions are markedly reduced by anticholinergic drugs. Gastric secretions are also decreased,but larger doses are necessary. Decreased intestinal motility and peristalsis prolong gastric emptying time. Lower esophageal sphincter pressure is reduced. Overall, the anticholinergic drugs do not prevent aspiration pneumonia. Eye Anticholinergics cause mydriasis (pupillary dilation) and cycloplegia (an inabiliy to accommodate to near vision); acute angle-closure glaucoma is unlikely following systemic administration of most anticholinergic drugs. Genitourinar y Anticholinergics may decrease ureter and bladder tone as a result of smooth muscle relaxation and lead to urinary retention, particularly in elderly men with prostatic hypertrophy Thermoregul ation Inhibition of sweat glands may lead to a rise in body temperature (atropine fever). Pharmacological properties
- 56. Pharmacokinetic
- 58. • Atropine sulfate is available in a multitude of concentrations • Dosage: As a premedication, atropine is administered IV OR IM in a range of 0.01–0.02 mg/kg, up to the usual adult dose of 0.4–0.6 mg. • Larger intravenous doses up to 2 mg may be required to completely block the cardiac vagal nerves in treating severe bradycardia. ATROPINE
- 59. • Atropine has particularly potent effects on the heart and bronchial smooth muscle and is the most efficacious anticholinergic for treating bradyarrhythmias. • A derivative of atropine, ipratropium bromide, is available in a metered-dose inhaler for the treatment of bronchospasm. • Ipratropiumsolution (0.5 mg in 2.5 mL) seems to be particularly effective in the treatment of acute chronic obstructive pulmonary disease when combined with a β- agonist drug (eg, albuterol). • The central nervous system effects of atropine are minimal after the usual doses, even though this tertiary amine can rapidlycross the blood–brain barrier. • Atropine has been associated with mild postoperative memory deficits, and toxic doses are usually associated with excitatory reactions. • An intramuscular dose of 0.01–0.02 mg/kg reliably provides an antisialagogue effect. • Atropine should be used cautiously in patients with narrow-angle glaucoma, prostatic hypertrophy, or bladder-neck obstruction .
- 60. • It is usually given IV • Solutions 0.3, 0.4, and 1 mg/mL • IV dose 5-10mcg • The lipid solubility allows transdermal absorption, and transdermal scopolamine has been used to prevent postoperative nausea and vomiting SCOPOLAMINE (Scopolamine hydrobromide )
- 61. • Scopolamine is a more potent antisialagogue than atropine and causes greater central nervous system effects. • Clinical dosages usually result in drowsiness and amnesia, although restlessness, dizziness, and delirium are possible. • The sedative effects may be desirable for premedication but can interfere with awakening following short procedures.
- 62. • Solution - 0.2 mg/mL. • The usual dose of glycopyrrolate is one-half that of atropine. • Premedication dose is 0.005–0.01 mg/kg up to 0.2–0.3 mg in adults. • Glycopyrrolate has a longer duration of action than atropine (2–4 h vs 30 min after IV) GLYCOPYRROLATE
- 63. • Almost devoid of central nervous system and ophthalmic activity . • Potent inhibition of salivary gland and respiratory tract secretions is the primary rationale for using glycopyrrolate as a premedication. • Heart rate usually increases after intravenous but not intramuscular administration.
- 64. The neuromuscular function of all patients receiving intermediate- or long- acting neuromuscular blocking agents should be monitored. In addition, peripheral nerve stimulation is helpful in assessing paralysis during rapid- sequence inductions or during continuous infusions of short-acting agents. The response of the nerve to electrical stimulation depends on three factors: • The current applied • The duration of the current • The position of electrodes. Monitoring Neuromuscular Blockade
- 65. 1. Visual and tactile evaluation is the easiest and least expensive way to assess the response to electrical stimulation applied to a peripheral nerve. The disadvantage of this technique is the subjective nature of its interpretation (present or absent, weak or strong). 2. Measurement of force using a force transducer provides accurate assessment (quantitative or objective) of the response elicited by electrical stimulation of a peripheral nerve. 3. Electromyography measures the electrical rather than mechanical response of the skeletal muscle. 4. Accelerometry devices are usually attached to the thumb, and a digital readout is obtained. The use of accelerometry is helpful in the diagnosis of residual paralysis. Recording the Response
- 66. Muscles do not respond in a uniform fashion to NMBDs. (There are differences in time to onset, maximum blockade, and duration of action.) • Muscles Surrounding the Eye. There seem to be important differences in the responses of muscles innervated by the facial nerve (stimulated 2–3 cm posterior to the lateral border of the orbit) around the eye. a. The response of the orbicularis oculi over the eyelid is similar to that of the adductor pollicis. b. The response the eyebrow (corrugator supercilii) parallels the response of the laryngeal adductors. (Onset is more rapid and recovery is sooner than at the adductor pollicis.) This response is useful for predicting intubating conditions. Choice of Muscle
- 67. • The adductor pollicis supplied by the ulnar nerve is the most common skeletal muscle monitored clinically. This muscle is relatively sensitive to nondepolarizing muscle relaxants, and during recovery, it is blocked more than some respiratory muscles such as the diaphragm and laryngeal adductors.
- 69. MONITORING NONDEPOLARIZING NEUROMUSCULAR BLOCKADE Single Twitch (intervals of >10 sec) Evaluate onset time (INTUBATION) Tetanus (frequency ≥30 Hz) Peak response followed by fade More sensitive than single twitch in detecting residual neuromuscular blockade Train-of-Four Ratio Height of fourth twitch to that of the first twitch More sensitive than single twitch and similar sensitivity to tetanic stimulation Evaluate responses manually or visually Posttetanic Count Applies when there is no response to single twitch, tetanus, or train-of-four stimulation Double-Burst Suppression Two short tetanic stimulations Evaluates the ratio of the second to the first response (correlates with the train-of-four ratio but is easier to detect manually)
- 73. Interpretation of monitoring depends on the context during which NMBDs or reversal drugs are given: • Monitorig onset-Predict intubating conditions • Monitoring surgical relaxation -Provide relaxation during surgical procedure • Monitoring recovery -Readiness for and effectiveness of reversal agents Clinical application
- 80. 1. file:///D:/My%20lessons/Anasthesiology%20Residency%201st/Resident%20Ankhzaya's%20files/Books %20of%20Anesthesiology/Barash's%20Handbook%20of%20Clinical%20Anesthesia%207th%20Edition .pdf 2. http://www.e-safe-anaesthesia.org/e_library/03/Suxamethonium_Update_1992.pdf 3. file:///C:/Users/enkhbat/Documents/Resident%20Ankhzaya's%20files/E-safe-Anaesthesia.org/E- Library/Basic%20science- Pharmacology/Pharmacology_of_neuromuscular_blocking_drugs_CEACCP_2004.pdf 4. file:///C:/Users/enkhbat/Documents/Resident%20Ankhzaya's%20files/E-safe-Anaesthesia.org/E- Library/Basic%20science- Pharmacology/Anticholinesterases_and_anticholinergic_drugs_CEACCP_2004.pdf 5. Morgan 5th Edition REFERENCES:
