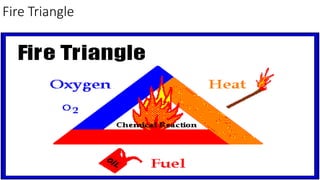
Fire Triange
- 2. Fuel • Fuel (solid, liquid, gaseous) must be present in certain concentrations. • Other sources of fuel source: if there is a leak, during filling operations, transfer operations, or excessive dusts. • Although we often cannot always eliminate these sources we can help by having good ventilation to keep vapors from building up. • Out door operations
- 3. Oxidizer • Oxygen is the most common oxidizer, especially that found in ambient air. • For oxygen, we often use “inerting” with nitrogen, helium blankets over flammable materials to reduce O2 content below that where you can have combustion.
- 4. Ignition Source • Heat is a common ignition source. • “Ignition sources are free!!!” • Although we can eliminate ignition sources, it is almost inevitable that an ignition source will be available if there is a large release of flammable material that cannot be diluted quickly.
- 5. Fire Tetrahedron • The fire tetrahedron or fire pyramid adds a fourth component—chemical chain reaction—as a necessity in the prevention and control of fires. • The free radicals formed during combustion are important intermediates in the initiation and propagation of the combustion reaction.
- 6. 3 T’s for Combustion Combustion efficiency can explained in terms of the three T’s; 1. Temperature: high enough to ignite and maintain ignition of the fuel (flash point) 2. Turbulence: or intimate mixing of the fuel and oxygen 3. Time: sufficient for complete combustion
- 7. What is a flame? A flame is a self-sustaining propagation of a localized combustion zone at subsonic velocities • Flame must be confined: flame occupies only a small portion of combustible mixture at any one time (in contrast to a reaction which occurs uniformly throughout a vessel) • A discrete combustion wave that travels subsonically is called a deflagration • Combustion wave may be also travel at supersonic velocities, called detonation • Fundamental propagation mechanism is different in deflagrations and detonations Laminar vs. Turbulent Flames: both have same type of physical process and many turbulent flame theories are based on an underlying laminar flame structure
- 8. Description of a Candle Flame The flame surface is where vaporized fuel and oxygen mix, forming a stoichiometric mixture. At the flame surface, combustion leads to high temperatures that sustain the flame. The elements of the process are: • Heat from the flame melts wax at the base of the candle flame. • Liquid wax moves upward by capillary action, through the wick towards the flame. • Heat from the flame vaporizes the liquid wax. • Wax vapors migrate toward the flame surface, breaking down into smaller hydrocarbons. • Ambient oxygen migrates toward the flame surface by diffusion and convection.
- 10. COMBUSTION PHENOMENA Selected Mechanisms of Flame Formation, Propagation, and Extinction, pp17 JOZEF JAROSINSKI BERNARD VEYSSIERE
- 12. Structure of a Candle Flame • In the outer zone complete combustion of the fuel takes place and the color of the flame is blue and is the hottest part of the flame. It is the non luminous part of the flame. • In the middle zone partial combustion of the fuel takes place and the color of the flame is yellow and is moderately hot part of the flame. It is the luminous part of the flame. • In the inner zone there are un-burnt vapors of the fuel and the color is black and is least hot part.
- 13. Heat and Mass Transfer Pathways
- 14. Radical Mechanism of Combustion The combustion can be explained by radical mechanism usually consisting of initiation, propagation, branching and termination. M is a third body in the system, which is non-reacting.
- 15. General Fuel Terms • Combustion – a chemical reaction in which a substance combines with an oxidizer and releases energy. • Explosion – rapid expansion of gases resulting in a rapid moving pressure or shock wave. • Mechanical Explosion – due to failure of vessel with high pressure non reactive gas • Detonation – explosion (chemical reaction) with shock wave greater than speed of sound • Deflagration – explosion (chemical reaction) with shock wave less than speed of sound
- 16. Flammability • Flash Point (FP) – a property of material used to determine the fire and explosive hazard. The lowest temperature of a liquid at which it gives off enough vapor to form an ignitable mixture with air. • Needs to be determined experimentally. • Different methods to determine, open cup and closed cup. Open cup is usually a few degrees higher
- 17. Flammability limits • Flammability limits are specify lean and rich fuel-oxidizer mixture ratio beyond which no flame will propagate • There is usually a range of compositions of a flammable vapor and air where combustion occurs. • Too little fuel (lean mixture) not enough fuel to burn. • Too much fuel (rich mixture) not enough oxygen to burn
- 18. Mechanism of Combustion • mechanism of combustion follows the complex phenomena with several elementary steps. ‘ • In the preliminary study, the possible steps on the overall reaction are to be assumed. • Large number of elementary reaction steps involving large number of intermediates are possible. • For example, a large number of species are involved in the combustion of simple hydrocarbons like CH4.
Editor's Notes
- Fundamentals of Combustion Processes by Sara McAllister l Jyh-Yuan Chen A. Carlos Fernandez-Pello, p 140
- COMBUSTION PHENOMENA Selected Mechanisms of Flame Formation, Propagation, and Extinction JOZEF JAROSINSKI BERNARD VEYSSIERE Pp 172