Global Site Contract Cycle Time Expectations by Country
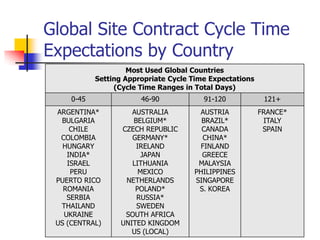
- 1. Global Site Contract Cycle Time Expectations by Country Most Used Global Countries Setting Appropriate Cycle Time Expectations (Cycle Time Ranges in Total Days) 0-45 46-90 91-120 121+ ARGENTINA* AUSTRALIA AUSTRIA FRANCE* BULGARIA BELGIUM* BRAZIL* ITALY CHILE CZECH REPUBLIC CANADA SPAIN COLOMBIA GERMANY* CHINA* HUNGARY IRELAND FINLAND INDIA* JAPAN GREECE ISRAEL LITHUANIA MALAYSIA PERU MEXICO PHILIPPINES PUERTO RICO NETHERLANDS SINGAPORE ROMANIA POLAND* S. KOREA SERBIA RUSSIA* THAILAND SWEDEN UKRAINE SOUTH AFRICA US (CENTRAL) UNITED KINGDOM US (LOCAL)
- 2. Country Experience with Site Contracting Russia, Poland Belgium Will accept CRO or sponsor template At least 3 agreements (Institution, Investigators, In Poland, need a final CTA before MOH submission Pharmacy) Brazil Will use CRO or sponsor template but will need to Multi-party CTAs/back and forth modify for the 3 parties Access to drug an issue France Argentina Prefers using own template CTAs usually not an issue Investigator and Institution have separate agreements Costs are low No reference to US law or FDA regulations China Germany CTAS not an issue Accept no templates Costs are low Any new invention belongs to the investigator MOH rate limiting Latin America India Easy to negotiate Will accept CRO or sponsor template Chili and Peru have two party agreements (PI and 2 party agreement (CRO and PI) – site does not sign CRO) Costs are low Hospitals refuse to sign agreement Things move quickly Clinical trial insurance not easy to obtain PIs don’t carry liability insurance
