Epivolve Manual
- 1. the group EPIVOLVE™ TRIPLE-DOSE EPINEPHRINE AUTO-INJECTOR AS DESCRIBED BY: Sonya Raab Jillian Hughes Stephen Ellias Nolan Flaherty
- 2. Rx Only Manufactured and Distributed by: the group The Nuvu Group Cambridge, MA 02139 USA © 2012 The Nuvu Group, Cambridge, MA. All rights reserved. This product may be covered by some or all of the following patents, patent applications and foreign equivalents thereof: U.S. Patent Nos. 5,358,489; 5,540,664; 5,665,071; and 7,297,136 and other pending U.S. Patent applications
- 3. the group EPIVOLVE™ TRIPLE-DOSE EPINEPHRINE AUTO- INJECTOR DEVICE COMPONENTS 2 CLINICAL PROBLEMS 3 THE EPIVOLVE™ SOLUTION 4 PROCEDURE 5 FDA CLASSIFICATION 6 IMPORTANT PRODUCT INFORMATION 7
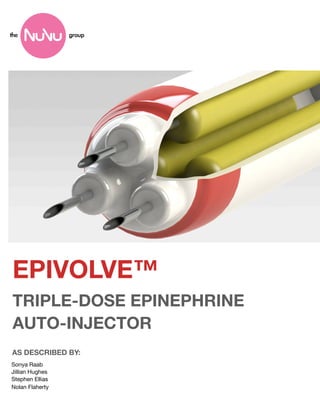
- 4. 2 EPIVOLVE™ Epinephrine Auto-Injector Device Components COMPONENTS AND DIMENSIONS 1 in ch es n ch i 4 1/ 4 Injection Dial Interchangeable Tri-Dose Epinephrine Cartridge ch Outer Customizable Shell in 2 1/ Needle DIMENSIONS Outer Unit: 4 1/4 in by 1 in Inner Cartridge: 3 in by 3/4 in Ejected Needle: 1/2 in
- 5. 3 EPIVOLVE™ Epinephrine Auto-Injector Clinical Problems CLINICAL PROBLEMS ANAPHYLAXIS A GROWING CONCERN Over the past two decades, anaphylaxis in children, teens and adults has risen dramatically, growing by 27% from 1998 to 2008 according to the CDC (graph right.) In the United States and Europe alone, estimates put the current population at risk for anaphylaxis at close to 100 million. EPI-PENS Rise in hospital discharges related to anaphylactic reactions from 1996 to 2006¹. A LACK OF ATTRACTION 5 out of 6 fatal anaphylactic reactions occur outside of one’s home and lack a dose of epinephrine. In a national survey, 87% of anaphylaxis victims admitted they choose to not epinephrine simply because they are embarrassed to be carrying around the device.² Because 1 in 2 anaphylaxis victims will need a second injection before medical attention arrives, it requires multiple devices to be carried at once. Devices on the market today offering more than one injection per device are difficult to use and require steps that may be difficult for a person in Epinephrine devices are lot to carry around on a daily basis. anaphylactic shock to complete. 1. Center for Disease Control/National Health Interview Survey, September 2008. 2. American Academy of Allergy Asthma and Immunology Survey, June 2005.
- 6. 4 EPIVOLVE™ Epinephrine Auto-Injector The Epivolve™ Solution EPIVOLVE™ VS. COMPETITION EPIVOLVE™ EPI-PEN™ TWINJECT™ Dosages per unit 3 1 2 Interchangeable ✔ Cartridges 4 1/4 inches by 5 1/2 inches by 6 inches by Unit Size 1 inch 3/4 inch 1/2 inch Customizable ✔ Enclosure Retractable Needle ✔ ✔ FDA Class II II II Cost Per Injection $26 $58 $39 WHAT MAKES EPIVOLVE™ DIFFERENT? INTERCHANGEABLE THREE DOSE CUSTOMIZABLE ENCLOSURE SLEEK DESIGN CARTRIDGE Epivolve™ allows its user to create customized The days of carrying a bulky auto-injector are Our patented triple dose cartridge supplies shells through our online website, giving users over. Epivolve™ features a lighter, smaller three measured does of epinephrine in case the freedom to create an Epivolve™ all their frame than its competition. Because it’s no multiple injections are necessary. Unlike other own. No longer will people feel embarrassed to bigger than a ballpoint pen, the device can be systems, the Epivolve™ requires only a simple carry around a bulky, plastic device. brought anywhere with little discomfort or dial turn to ready a new dose. And when all embarrassment. three are used, the cartridge can be simply removed and refilled by your doctor.
- 7. 5 EPIVOLVE™ Epinephrine Auto-Injector Procedure PROCEDURE STEP 1 STEP 2 STEP 3 REMOVE EPIVOLVE™ FROM CASE PULL BLUE TAB GRIP EPIVOLVE FIRMLY IN HAND Remove the Epivolve from its accompanying At the top of the Epivolve, there is a blue tab Grip the Epivolve firmly in hand with the orange case. If no case, skip this step. that must be pulled before injection. No not end pointing towards your body. touch the orange end. STEP 4 STEP 5 THRUST DEVICE AT RIGHT ANGLE INTO SEEK MEDICAL ATTENTION THIGH. HOLD FOR 5 SECONDS. Medical attention is still necesary immediately Thrust your Epivolve into your upper thigh and following injection. Dial 911 or have another hold in for 5 seconds to let medicine inject. person take you to a hospital IMMEDIATELY. If After five seconds, remove from leg and turn another anaphylactic shock is to come on knob at top of device. again, repeat steps for another injection.
- 8. 6 EPIVOLVE™ Epinephrine Auto-Injector FDA Classification FDA CLASSIFICATION CLASSIFICATION Epivolve™ has been registered as a Class II medical device by the Food and Drug Administration. The device has a mild risk for side effects (listed below) and requires users to seek contact of medical help IMMEDIATELY after injection. INDICATION Epivolve™ (epinephrine injection, USP 1:1000) is indicated in the emergency treatment of severe allergic reactions (Type I) including anaphylaxis to stinging insects (e.g. order Hymenoptera, which includes bees, wasps, hornets, yellow jackets and fire ants), and biting insects (e.g. triatoma, mosquitoes), allergen immunotherapy, foods, drugs, diagnostic testing substances (e.g. radiocontrast media), and other allergens, as well as anaphylaxis to unknown substances (idiopathic anaphylaxis) or exercise-induced anaphylaxis. SAFETY INFORMATION Epivolve™ should only be injected into the anterolateral aspect of the thigh. Accidental injection into the hands or feet may result in loss of blood flow to the affected area and should be avoided. DO NOT INJECT INTO BUTTOCK. DO NOT INJECT INTRAVENOUSLY. Epinephrine should be administered with caution to patients with cardiac arrhythmias, coronary artery or organic heart disease, or hypertension. In patients with coronary insufficiency or ischemic heart disease, epinephrine may precipitate or aggravate angina pectoris as well as produce ventricular arrhythmias. It should be recognized that the presence of these conditions is not a contraindication to epinephrine administration in an acute life-threatening situation. Adverse reactions to epinephrine may include flushing; apprehension; syncope; tachycardia; thready pulse with a fall in blood pressure; convulsions; respiratory difficulties; vomiting, diarrhea or cramps; pruritus; or angioderma. Epivolve™ is designed as an emergency supportive therapy only and is not a replacement or substitute for immediate medical care. Since the doses of epinephrine delivered from Epivolve™ are fixed, the physician should consider other forms of injectable epinephrine if doses lower than those available from Epivolve™ are felt to be necessary (e.g., patients who weigh less than 15 kilograms approximately 33 pounds). IN ALL CASES, THE PHYSICIAN SHOULD INSTRUCT THE PATIENT AND/OR ANY OTHER PERSON WHO MIGHT BE IN A POSITION TO ADMINISTER THE EPINEPHRINE, IN THE PROPER USE OF EPIVOLVE™.
- 9. 7 EPIVOLVE™ Epinephrine Auto-Injector Important Product Information IMPORTANT PRODUCT INFORMATION WHO SHOULD NOT USE EPIVOLVE™? There are no absolute contraindications to the use of Epivolve™ in a life-threatening allergic reaction. People with certain medical conditions have a higher chance of getting serious side effects from Epivolve™. Tell your doctor about all your medical conditions, but especially if you: • have heart disease or high blood pressure • have diabetes • have thyroid conditions • have asthma • have depression or other mental disease • have Parkinson’s disease • are pregnant • are allergic to any of the ingredients in Epivolve™ Tell your doctor about all the medicines you take, including prescription and non-prescription medicines, vitamins, and herbal supplements. Some medicines may cause serious side effects if taken while you use Epivolve™. Some medicines may affect how Epivolve™ works, or Epivolve™ may affect how your other medicines work. Diabetic patients may need to adjust the dose of their diabetes medicines or insulin after using Epivolve™. STORAGE The medicine in Epivolve™ can be damaged by light. Therefore, keep it in an area of general low light. Keep it at room temperature and protect it from freezing. Do not refrigerate. Keep Epivolve™ with you at all times. Check your Epivolve™ regularly to be sure: • it has not expired • the medicine in Epivolve™ is not cloudy, discolored, or has particles in it. Replace the Epivolve™ if needed.
- 10. 8 EPIVOLVE™ Epinephrine Auto-Injector Important Product Information SIDE EFFECTS Overdosage can cause dangerously high blood pressure, stroke, or death. If you take certain medicines, you may develop serious life-threatening side effects from the epinephrine in Epivolve™. Be sure to tell your doctor about all the medicines you take, especially medicines for asthma. Patients with certain medical conditions, or who take certain medicines, may get more side effects from Epivolve™, or the side effects may last longer. This includes patients who take certain types of medicines for asthma, allergies, depression, hyperthyroidism, high blood pressure, and heart disease. Patients with heart disease may feel chest pain (angina). Patients with mental disease or Parkinson’s disease may have worsening symptoms of their illness. Epivolve™ (epinephrine) can cause the following reactions. Some reactions can be serious. They usually go away with rest. Common side effects include, but not limited to: • fast, irregular heartbeat • throbbing headache • paleness • feelings of over excitement, anxiety, or fear • weakness or shakiness • dizziness • nausea and vomiting
