Espco plecture jpmbrugge2015
- 1. See discussions, stats, and author profiles for this publication at: https://www.researchgate.net/publication/293639908 Who, when why Opioid free anesthesia. Overview of actual use of OFA worldwide. Presentation · December 2015 DOI: 10.13140/RG.2.1.3068.6482 CITATIONS 0 READS 10,054 1 author: Some of the authors of this publication are also working on these related projects: opioid free anesthesia View project deep NMB achieving, reversing and monitoring View project Jan Paul Mulier AZ Sint-Jan Brugge-Oostende 178 PUBLICATIONS 1,287 CITATIONS SEE PROFILE All content following this page was uploaded by Jan Paul Mulier on 09 February 2016. The user has requested enhancement of the downloaded file.
- 2. OFA Brugge 19 Dec 2015 Jan Paul Mulier Departement of Anesthesiology AZ Sint – Jan AV Brugge Belgium Mulier ESPCOP Brugge 2015 1 Opioid Free Anaesthesia Who?, When?, Why?, Pos?, Neg?, How? 1150 2015
- 3. Patient 2 h post surgery on Ward Mulier ESPCOP Brugge 2015 2 No nausea No pain No cold Comfortable Sufficient awake
- 4. Overview 1. Who is giving OFA today? survey – Worldwide, more positive than negative responses 2. Protocols used to achieve OFA today 3. What is the outcome effect of OFA vs OA? 1. Advantages and disadvanges of OFA from survey 2. 50 pat Randomised bariatric surgery • It is possible, equal hemodyn stable, less post op analgetics, no cortisol rise, better VAS score 3. 500 pat Prospective Qo40 quality of recovery bariatric surgery • ERAS and improvement patient satisfaction 4. 5000 pat Retrospective lap gastric bypass • Less major complications Mulier ESPCOP Brugge 2015 3
- 5. Do you give general anaesthesia without opioids (OFA) (in the absence of regional anaesthesia) today? Mulier ESPCOP Brugge 2015 4 6% 18% 39% 28% 9% Do you give OFA today? Yes for most patients except when contra indications. Yes for a selected group of patients. No but reduce opioids (LOA) for some or all patients. No but willing to learn more about it. No and don’t see the possible advantages. 623 Answers on survey with 21 questions Anaesthesiologist who visited Bruges since the OFA introduction in 2011 (307 sent, 89 answers), Most Belgian anaesthesiologists (2350 sent, 392 answers) Some anaesthesiologists worldwide (823 sent, 117 answers) Web based link on ESPCOP site (25 answers)
- 6. Response from 623 anaesthesiologists Mulier ESPCOP Brugge 2015 5 12% 12% 15%61% Your work experience. I am in training Specialist Anaesthesiologist for 0 – 5 years Specialist Anaesthesiologist for 6 – 10 years Specialist Anaesthesiologist for more than 10 years 37% 34% 29% Where do you work? Public or private hospital with no academic activity or research Public or private hospital with academic activity or research University hospital
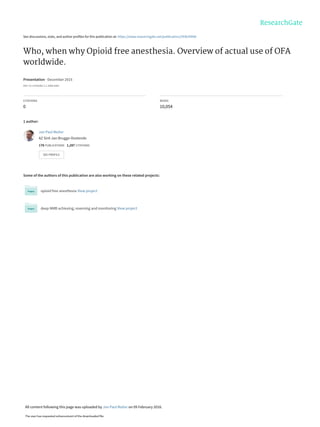
- 7. OFA is used in 26 countries Mulier ESPCOP Brugge 2015 6 Saved from: http://www.amcharts.com/visited_countries/ Belgium Dutch speaking 56 Belgium French speaking 40 Mexico 4 Nigeria 4 Switserland 4 United Kingdom 4 Brasil 3 Russia 3 South Africa 3 USA 3 Austria 2 Netherlands 2 Poland 2 Algeria 1 Argentina 1 Canada 1 China 1 Denmark 1 Egypt 1 France 1 Greece 1 Israel 1 Malaysia 1 New Zealand 1 Saudi Arabia 1 Turky 1 Missing country 6
- 8. How long have you been giving OFA? Mulier ESPCOP Brugge 2015 7 23% 32% 20% 25% For. how long have you performed this opioid free (OFA) method? Started this year More than 1 year More than 2 year More than 5 year 33% 37% 30% Work place of OFA users Public or private hospital with academic activity or research Public or private hospital with no academic activity or research University hospital 12% 7% 17% 64% Workexperience of OFA users I am in training Specialist Anaesthesiologist for 0 – 5 years Specialist Anaesthesiologist for 6 – 10 years Specialist Anaesthesiologist for more than 10 years
- 9. Patients selected for OFA. (all that apply) Mulier ESPCOP Brugge 2015 8 0% 20% 40% 60% 80% 100% Indicate which patients you select for OFA and LOA OFA LOA Other: high PONV risk pregnant or breastfeeding free flap surgery combined with locoregional. elderly chronic pain patients knee and hip to accellerate mobilisation Respiratory concerns is the reason today to avoid opioids
- 10. Main reasons why you reduce or avoid opioids peri-operatively. Mulier ESPCOP Brugge 2015 9 0% 20% 40% 60% 80%
- 11. What methods do you use to reduce (LOA) or avoid (OFA) opioids peri-operatively? Mulier ESPCOP Brugge 2015 10 0% 20% 40% 60% 80% 100%
- 12. When you use additives for general anaesthesia what do you add (all that apply) Mulier ESPCOP Brugge 2015 11 0% 20% 40% 60% 80% 100%
- 13. Ketamine dose (59 answers) • At induction 0,1 to 2 mg/kg (59 answers) – Mean 0,4 to 0,6 mg/kg – Mediaan 0,5 mg/kg • Maintenance 0,15 to 2 mg/kg/h (10 answers) – Mean 0,3 mg/kg/h – Mediaan 0,25 mg/kg/h • Large variation: Why? (1-20) – Preventing tolerance (0,2 mg/kg) (hyperalgesia) to analgesia (0,5 mg/kg) or anesthesia (2 mg/kg). (hallucinations) Mulier ESPCOP Brugge 2015 12
- 14. Lidocaine dose (34 answers) • At induction 0,6 to 2 mg/kg (59 answers) – Mean 1,2 - 1,6 mg/kg/h – Mediaan 1 - 1,5 mg/kg/h • Maintenance 1 to 5 mg/kg/h (10 answers) – Mean 1,25 - 2 mg/kg/h – Mediaan 1 - 1,5 mg/kg/h • Variation: Why? (1-4) – Minimum 1 mg/kg required – > 1,5 mg/kg or infusion for long procedures Mulier ESPCOP Brugge 2015 13
- 15. Clonidine dose (50 answers) • At induction 0,5 to 4 mkg/kg (50 answers) – Mean 1,6 – 2,5 mkg/kg – Mediaan 2 mkg/kg • Maintenance 0,3 to 0,6 mkg/kg/h (10 answers) – Mean 0,3 mkg/kg/h • Post operative ? – Bolus 2 x 1 – 2 mkg/kg • Large variation: Why? (1-8) – From additive (0,5) to strong sympathetic blocker (4) – Bradycardia, hypotension does not increase with dose – Prolonged sedation if > 2 used for short procedure Mulier ESPCOP Brugge 2015 14
- 16. Dexmedetomidine dose (11 answers) • At induction 0,1 to 1,4 mkg/kg – Mean 0,7 – 0,9 mkg/kg/h – Mediaan 0,7 mkg/kg/h • Maintenance 0,2 to 1 mkg/kg/h – Mean 0,3 mkg/kg/h • Post operative ? – 0,1 – 0,2 mkg/kg/h • Larger variation than clonidine: Why? (1-14) – From additive (0,1) to strong sympathetic blocker (1) – Bradycardia, hypotension does not increase with dose – Prolonged sedation if > 1 used for short procedure Mulier ESPCOP Brugge 2015 15
- 17. Problems with OFA Mulier ESPCOP Brugge 2015 16 0% 20% 40% 60% 80% What do you do when a hypertensive/tachycardia reaction happens during OFA? 0% 20% 40% 60% 80% 100% What do you do when a hypotension/ bradycardia reaction happens during OFA? (all that apply) 87% 7% 6% Have you had any serious adverse events when reducing opioids (LOA) or working opioid free (OFA)? Not until now Yes but probably not related to LOA/OFA Yes and probably related to LOA/OFA 0 0,2 0,4 0,6 0,8 What do you do when patient has immediately post operative pain.
- 18. Pro & Contra OFA - LOA Mulier ESPCOP Brugge 2015 17 0% 20% 40% 60% 80% 100% Disadvantages of LOA OFA 0% 20% 40% 60% 80% 100% Advantages of LOA OFA
- 19. Obstacles to adopt OFA? Mulier ESPCOP Brugge 2015 18 0% 20% 40% 60% 80% 100% Insufficient guidelines. No training Outcome not proven yet. Other limited data. Not aware of OFA. Expensive. outside prescription label. More work.
- 20. Opioid free anaesthesia (OFA) in Bruges started in 2010 - 2011 • Randomized study, ready for publication on 50 lap gastric bypass patients getting OFA vs. OA. – Comparing post operative morphine PCIA • Prospective study on 500 lap gastric bypass patients getting OFA vs. OA. Thesis M Crivits 2014 – Comparing ERAS evaluation using the Qo40 questionnaire • Retrospective analysis 5000 lap gastric bypass patients getting OFA vs. OA – Comparing complications and outcome. Mulier ESPCOP Brugge 2015 19
- 21. • No demographic differences between the two groups with regard to age, weight, length, BMI, gender, the incidence of pre operative OSAS. • No difference in Kalkman score (1. prediction of severe postoperative pain) and in the Amsterdam Preoperative Anxiety and Information Scale (2. APAIS). • No differences in hemodynamic problems per operative, like bradycardia, hypotension, tachycardia or hypertension defined as a change of 20 % with or without a treatment requirement. • Kalkman score and APAIS score did not predict morphine use or pain score. VAS scores are better with a lower morphine consumption post operative Mulier ESPCOP Brugge 2015 20 2013 R. Wouters; J. P. Mulier Randomized study in 50 lap RNY patients Patients got OFA or Opioids per-op only Same post op drugs: paracetamol and diclofenac Morphine pump PCIA: VAS score at PACU and at ward first day
- 22. Randomized study on 50 pt Mulier ESPCOP Brugge 2015 21 Table 1: Postoperative at PACU (Nr of pat) OA (21) OFA (23) p value low saturation with O2 mask 8 0 <0.001 z score obstructive breathing 3 0 0.06 z score PONV 7 3 <0.001 z score shivering or having cold 5 0 0.013 z score VAS score (mean) 4,88 1,66 <0.001 t test Morphine need (mean in mg) 15,3 4,91 0.004 t test Table 2: Postoperative at Ward OA (21) OFA (23) p value emotional state (9) 7,22 8,11 0.051 z score physical comfort (12) 7,94 10,39 <0.001 z score psyche support (7) 6,60 6,73 0.62 z score physical independence (5) 3,80 4,71 0.007 z score pain score (7) 4,50 6,11 <0.001 z score sleep score (4) 2,11 3,30 0.040 z score total Qo40 score (40) 29,60 35,81 <0.001 z score cortisol change mg 10,55 3,57 0.029 t test VAS score (mean) 3,29 2,04 0.016 t test Morphine need (mean in mg) 18,18 14,73 0.330 t test 0,00 2,00 4,00 6,00 8,00 10,00 12,00 14,00 16,00 18,00 20,00 paza 30 min paza 60 min paza 120 min total morfine mg VASmgmorfine Vas score and morfine at PAZA OA OFA 0,00 5,00 10,00 15,00 20,00 25,00 30,00 35,00 40,00 cortisol before anesthesia cortisol after anestehsia cortisol plasma level cortisol plasma levels OA OFA M+ Pain VAS
- 23. 500 patients questioned day1 post lap RNY OFA vs. OA Mulier ESPCOP Brugge 2015 22 0% 10% 20% 30% 40% 50% 60% 70% 80% 90% 100% percentageofpositveanswers Qo40 Questions that are significant different OFA versus OA OFA OA * Chi square p < 0,05 * * * * ** * * * * *
- 24. Retrospective study using all consecutive lap RNY (from March 1st 2011 to June 31st 2015: 5061 patients ). B Dillemans – J Mulier Bariatric Center Bruges • A prospectively kept database of all the patients undergoing fully stapled laparoscopic Roux-en-Y gastric bypass (FS-LRYGB) in a standardized fashion by a single surgeon or under his direct supervision since May 2004 contains today more than 10 000 consecutive procedures. • Low Opioid anesthesia (LOA) by using clonidine started in 2010. In 2011 total opioid free anesthesia (OFA) was given, first by using clonidine and from 2012 by using dexmedetomidine. Around half of the patients were selected for opioid free anesthesia (OFA) from the beginning, based on the attending supervising anesthesiologist and his assumed risk of using opioids. • A small group of patients were still given a low opioid anesthesia (LOA) by adding only a limited amount of additives in an effort to learn how to handle the switch to total opioid free anesthesia. • Patients given OA: 2451 OFA: 2337 LOA: 264 missing info: 9 Mulier ESPCOP Brugge 2015 23
- 25. All one month follow-up complications after lap RNY are recorded. Mulier ESPCOP Brugge 2015 24 2409 2276 248 83 61 16 0 500 1000 1500 2000 2500 3000 OA OFA LOA Number of complications in Lap RNY 2011-2015 complications no complications typeanesthesie numberofprocedures Nonsurgical complications percentage bleedingcomplications percentage surgeryrelatedwithout bleedingcomplications percentage OA 2492 40 1,61% 33 1,32% 5 0,20% OFA 2337 21 0,90% 24 1,03% 12 0,51% LOA 264 9 3,41% 3 1,14% 2 0,76% total 5093 70 1,37% 60 1,18% 19 0,37% p = 0.043 * * logis'c regression coef p value OFA -0,392 0,037 age 0,026 0,001 gender: male 0,413 0,031 Re interven'on 0,634 0,003 OSAS 0,23 0,222 OSAS in OA group 0,001 OSAS in OFA group 0,196 BMI 0,009 0,321 Risk for complications decreases by OFA increases by age, male gender, re intervention BMI no effect, OSAS increases risk in OA
- 26. Complications type after OA OFA LOA Mulier ESPCOP Brugge 2015 25 0 5 10 15 20 25 30 35 40 45 LOA OFA OA OFA has more surgery related complications OA has more respiratory, infectious and bleeding related complications LOA has more complications on average but group is small
- 27. Are the groups comparable? • Not randomized prospective database. • Variable OA OFA LOA p • Male 28,21% 28,07% 24,62% 0,51 • BMI 40,89 41,18 41,66 0,41 • Age 41,09 42,19 * 41,27 0,009 • OSAS 37% 39% * 31% * 0,024 • Redo 15% 19% * 16% 0,001 • Not comparable: OFA has more patients with OSAS and redo surgery. • OSAS if STOPbang > 3, probably underscored by missing data Mulier ESPCOP Brugge 2015 26
- 28. Are other outcome variables different? Not randomized prospective database. • Variable OA OFA LOA p • Unplanned ICU 0.56% (14) 0.3% (4) 0.38% (1) 0.38 • No of hospital days 2.73 2.53 * 2.86 0.001 • 1 week revisions 0.36% (9) 0.13 (3) 0.38 (1) 0.10 • Hospital readmission 0.88% (22) 1.03% (24) 1.89% (5) 0.29 • Morphine day 0 (SD) 21 mg (0.99) 6 mg (0.48)* 15 mg (1.9)* 0.001 • No no-opioids postop 1.36% (34) 8.7% (205) 5.6% (15) 0.001 • OFA patients have a shorter hospitalization and need less or no opioids on the first day. Mulier ESPCOP Brugge 2015 27
- 29. Mulier ESPCOP Brugge 2015 28 CURRENT OPINION Perioperative opioids aggravate obstructive breathing in sleep apnea syndrome: mechanisms and alternative anesthesia strategies Jan P. Mulier Purpose of review Opioids induce and increase the severity of most sleep-disordered breathing in all patients, but especially in morbidly obese patients. Discussed herein are the direct impact and mechanisms of opioids on inducing and exacerbating obstructive sleep apnea syndrome in normal and morbidly obese patients. Recent findings Respiratory depression is a larger problem than obstructive sleep apnea syndrome during the first night after an opioid anesthesia because of the reduced amount of deep sleep and rapid-eye-movement sleep. Acute tolerance to the analgesic effects of opioids can be observed after one anesthetic opioid dose, although tolerance to the side-effects of opioids develops more slowly. Therefore, it makes sense to avoid all opioids intraoperatively. A recently developed multimodal nonopioid anesthesia method may prevent development of acute tolerance and facilitate postoperative pain management with less opioids and sleep- disordered breathing. Summary A multimodal nonopioid anesthesia method avoids the necessity for intraoperative opioids, reduces the need for postoperative opioid use, and improves analgesia with less narcotic. Keywords analgesia, anesthesia, obstructive sleep apnea syndrome, opioids REVIEW 015 Wolters Kluwer Health, Inc. Unauthorized reproduction of this se patients, with or without OSAS, quent oxygen desaturation episodes Curr Opin Anesthesiol 2015, 28:000–000 DOI:10.1097/ACO.0000000000000281 ght ß 2015 Wolters Kluwer Health, Inc. All rights reserved. www.co-a
- 30. Acute morphine tolerance was known for a long time but forgotten? Mulier ESPCOP Brugge 2015 29 Br. J. Pharmac. Chemother. (1968), 33, 245-256. ACUTE TOLERANCE TO NARCOTIC ANALGESIC DRUGS IN RATS BY B. M. COX, M. GINSBURG AND 0. H. OSMAN From the Department of Pharmacology, Chelsea College of Science and Technology, London, S.W3 (Received November 2, 1967) It has been reported that treatment of rats and mice with actinomycin D, an antibiotic inhibitor of protein synthesis, diminishes the impairment of the analgesic effect of morphine normally observed during chronic treatment with the analgesic (Cohen, Keats, Krivoy & Ungar, 1965). The widespread metabolic disturbances that arise during prolonged treatment with inhibitors of protein synthesis (Young, Robinson & Sacktor, 1963) make it difficult to interpret this result, and in fact, some of the animals receiving higher doses of actinomycin died. In the experiments reported in the present paper it is shown that in rats, tolerance to morphine and other analgesics given by continuous intravenous infusion develops within 4 hr and this makes it possible to study the effects of actinomycin D on tolerance while overall metabolism is apparently unimpaired. METHODS Preparation of animals for intravenous infusion Male or female Wistar rats weighing between 130 and 250 g were anaesthetized with ether. A polyethylene cannula filled with saline was inserted into a jugular vein and the free end was exteriorized through a stab wound in the skin at the back of the neck. On the next day, solutions of drugs (details in RESULTS) dissolved in 0.9%1 (w/v) sodium chloride solution were infused into the conscious animals from a continuous infusion apparatus connected to the jugular vein cannulae by fine polyethylene tubes. The rate of infusion was 1 ml./hr. Measurement of analgesia Analgesia was measured by a modification of the method of Green & Young (1951). A force was applied to the tip of a rat's tail by the plunger of a syringe connected to a compressed gas supply (Morton, 1962). Pressure developed within the syringe (which was measured by a U-tube mercury 246 B. M. COX, M. GINSBURG and 0. H. OSMAN drug treatment, at times after drug administration, and the maximum pressure, respectively.) Pmax was taken arbitrarily at 36 cm Hg, but in practice pressure stimuli greater than 32 cm Hg were never used. For example, where an animal with PA=6 responded to a pressure of 32 cm Hg, the 32-6 analgesic index would be -6 =0.87; where such an animal did not respond at 32 cm Hg the index 36-6 was taken as 1.00. Motor co-ordination test The time during which rats (tested individually) were able to balance on a rotating horizontal rod was measured. The P.V.C. covered rod (diameter 25 mm.) was rotating at 1.1 rev/min. Nitrogen excretion Measurements of total nitrogen content of urine were made by Kjeldahl's method. The following drugs were used: morphine HC1, diamorphine HCI, pethidine HCI, etorphine HCl, actinomycin D (Dactinomycin, Merck, Sharp and Dohme). RESULTS Preliminary experiments were carried out to discover a suitable range of infusion rates of morphine into conscious rats. The criteria chosen were such that, at least initially, significant levels of analgesia should be produced and that the rates of infusion should 1.0 0.8 x -04 ._ C S us c do d 0.6 0.4 0.2 -0.2 F a ' ' ' 'I I 1 2 4 6 8 Time from start of infusion (hr) Fig. 1. Mean analgesic indices during intravenous infusions of morphine. 0, Morphine 5 mg/kg/hr (n=4); *, morphine 7.5 mg/kg/hr (n=7); A , morphine 10 mg/kg/hr (n=5); *, 0.9% sodium chloride solution (n=4). Morphine Tolerance starts after 2 hours No analgesic effect after 8 hours Br. J. Pharmac. Chemother. (1968), 33: 245
- 31. Remifentanyl tolerance in 90 min No analgesic effect after 3 h Mulier ESPCOP Brugge 2015 30 Rapid Development of Tolerance to Analgesia During Remifentanil Infusion in Humans H. Ronald Vinik, MD*, and Igor Kissin, MD, PhDt *Department of Anesthesiology, University of Alabama at Birmingham, Birmingham, Alabama; and tDepartment of Anesthesia, Harvard Medical School, Brigham and Women’s Hospital, Boston, Massachusetts Studies in experimental animals have demonstrated a rapidly developing acute tolerance to the analgesic ef- fect of opioids administered by continuous IV infusion. The aim of the present study was to determine whether acute tolerance plays an important role in the analgesic effect of remifentanil provided by IV infusion to hu- mans. The analgesic effect of remifentanil, infused at a constant rate of 0.1 pg *kg-’ * min-’ for 4 h, was evalu- ated by measuring pain tolerance with thermal (2°C water) and mechanical (pressure) noxious stimulations in 13 paid volunteers. The constant-rate infusion of remifentanil resulted in a threefold increase in pain tol- erance with both tests. After reaching its maximum in 60-90 min, the analgesic effect of remifentanil began to decline despite the constant-rate infusion, and after 3 h of infusion, it was only one fourth of the peak value. A comparative rate in the development of acute tolerance measured in terms of time to 50% recovery during infu- sion was 129 + 27 min (mean + SD) with the cold water test and 138 + 39 min with the pressure test. We con- clude that the development of tolerance should be in- cluded in the calculations for target-controlled infu- sions. Implications: Our study shows that tolerance to analgesia during remifentanil infusion is profound and develops very rapidly. The administration of opioids during anesthesia based on target-controlled infusions should include corrections for the development of tolerance. (Anesth Analg 1998;86:1307-11) S tudies of constant-rate IV infusions of opioids in dogs and rats have demonstrated a profound decrease in analgesia within 2-8 h after the be- ginning of the infusion (l-4). The analgesic effect of morphine declines profoundly, despite the absenceof a decrease in morphine brain concentration (3). This based on models of tolerance development. The aim of the present study was to determine whether acute tolerance plays an important role in the analgesic ef- fect of remifentanil administered by IV infusion to humans. usly applied to the tibia, and the volunteer was ked to report when the pain became intolerable. The ration of the volunteer’s tolerance to the pain stim- us was recorded. The pain stimulus was discontin- d after a maximum of 60 s. Pain in the tibia was ed immediately after cessation of the pressure (ver- l scale from 0 to 10). Sedation was assessedby the lunteers by estimating drowsiness on a scalefrom 0 10 (0 = alert, 10 = very sleepy). The testing session began with measurement of seline pain tolerance values. Pressure tests were rformed first, followed by cold water tests 5 min er. For the first 30 min, each volunteer received an usion of isotonic sodium chloride solution, fol- wed by repeated measurements of pain tolerance. he volunteer was blinded to the contents of the in- sion. If both cold and pressure tolerance increased >30%, the subject was excluded from the study, nd remifentanil was not administered. The aim of the otonic sodium chloride infusion was to eliminate acebo responders. Of the 13 volunteers, 2 were ex- uded becauseof the response to the isotonic sodium loride infusion. Immediately after the second pain tolerance meas- ement, remifentanil was infused at a constant rate of 1 pg * kg-r * min-i and maintained for 4 h. Meas- ements of all variables were performed every min for 2 h and hourly thereafter. Cold water and pressure test responseswere treated s continuous variables. The time to 50% recovery reached its peak in 60-90 min. After reaching a maximal analgesicresponse,analgesiabegan to decline in eight of nine subjectsdespite the continuing infusion of remifen- tanil (Fig. 1). In one such subject,the cold water test was tolerated beyond the cutoff limit (180s) until the end of remifentanil infusion. Therefore, the possibility for the development of tolerance to analgesiawas questionable. However, with the pressure test, the same subject dem- onstrated a decline in analgesiaafter the peak response to remifentanil infusion. After 3 h of remifentanil infu- sion, the analgesicresponseto the cold water testwas, on average, only one fourth of the peak value. Comparative degrees of the analgesia reduction during remifentanil ~~.:‘:11(1 ~1 2 IO- c” o- Remifentanil O.lpg.kg-‘.min-1 1 I I I I I I I I -30 0 30 60 90 120 150 180 210 240 Time (min) Figure 1. Time course of the analgesic effect of remifentanil (constant-rate infusion of 0.1 mg . kg-’ . min-I) as determined by using a cold water pain tolerance test. “P < 0.0001 versus Time 0; bP < 0.05 versus 90 min; ‘P < 0.0001 versus 90 min. Anesth Analg 1998;86:1307 Reason why after TIVA: • Crying of pain if no opioid is continued even when minor or not painfull surgery • Shivering and need for bair hugger • Vomiting requiring propofol and ondansetron
- 32. Have OSAS patients more complications after OA vs. OFA? Mulier ESPCOP Brugge 2015 31 • Not randomized prospective database. • Variable No OSAS OSAS p • OA 2.02% (31) 5.05% (44)* 0.001 – OFA 1.95% (27) 2.49% (22) 0.39 • LOA 4.62% (8) 5.19% (4) 0.67 • Under standard OA, OSAS patients have a significant higher complication rate. • Under OFA, OSAS patients have not a higher complications rate compared to non OSAS patients. • LOA has a larger complication rate, the group is smaller and shows a learning curve! • OFA is difficult in the beginning and requires an effort to learn.
- 33. OFA induction s Sympathetic block: 10 min before induction s Dexmedetomidine: 0,3 ug/kg IBW (20 -30 ug) further loading to 0,5 after intubation s Local anesthetics iv: 1 min before induction (hypnotic and rapid stress block) s Lidocaine: 1,5 mg/kg IBW (100 mg) s Hypnotics iv: induction (hypnotic) s Propofol 2,5 mg/kg IBW (200 mg) s Hemodynamic stabilization (cave preload reduction) s Mg Sulfate 40 mg/kg IBW (2,5 gr) s NMB if needed for anesthesia or surgery s Anti-inflammatory agents before surgery (lap) corticoiden, NSAID s Dexamethasone 10 mg, Diclofenac 75 - 150 mg s NMDA block (analgesic and blocking hyperalgesia by opioids) s Ketamine 10 - 25 mg bolus or slow infusion or at end operation s Have B blocker, Ca antagonist, ephedrine and phenylephrine ready s Metoprolaat 1-5 mg, Nicardipine 1-5 mg, Ephedrine 3-9 mg, Phenylephrine 10-30 ug Mulier ESPCOP Brugge 2015 32
- 34. OFA maintenance • Sympathetic block – Dexmedetomidine: 0,5 ug/kg/h. (long half life) stop when 1 ug/kg is reached – Or Clonidine 150 300 ug loading up (very long half life) • Local anesthetics – Procaine 0,1 % 2 mg/kg/h short half life and safer except allergic reactions – (or Lidocaine 1 % 1 – 3 mg/kg/h high dose has prolonged hypnotic effects) – Toxic dose lidocaine is probably very high > 10 mg/kg • Mg Sulfate 2,5 mg/kg IBW/h • Inhalation anesthesia – Sevoflurane, Desflurane 0,8 – 1 MAC with BIS around 40%. – Propofol infusion higher dose than TIVA required, difficult and BIS needed. • NMDA block (if opioids might get used post op) – Ketamine 50 mg over 12 h • Paracetamol 4 gr/24h post op Mulier ESPCOP Brugge 2015 33
- 35. Opioids are not generally accepted anymore! Balanced anesthesia: TIVA: Inhalation, opioids, NMB propofol, opioids, NMB 34 1. hypnosis 2. Sympathetic block 3. relaxation Unconsciousness Hemodynamic stability Immobility (relative) We don’t need analgetics to achieve hemodynamic stability Direct sympathetic block is possible. Mulier ESPCOP Brugge 2015
- 36. Conclusion Patients love OFA as ERAS improves, nurses see the difference on the ward Every change is difficult, requires more work and increases risks. You will never see the difference if you do follow your patient post-operative. Mulier ESPCOP Brugge 2015 35
- 37. LIVE OPIOID FREE ANAESTHESIA December 18th 2015 AZ Sint-Jan AV Ruddershove 10 Bruges, Belgium Flyer_Escop_6_18-12-2015.indd 1 19/05/15 20:02 December 19th 2015 Crowne Plaza, Burg 10 Bruges, Belgium 6th ESPCOP meeting Following the advice in using less peri-operative opioids in morbidly obese patients, it has now become time to discuss “from low-opioid to opioid free” anaesthesia (OFA). Why, how and when do we use low-opioid or opioid free anaesthesia combined with post-operative multimodal analgesia in morbidly obese patients? Flyer_Escop_6_19-12-2015.indd 1 19/05/15 20:03 Mulier ESPCOP Brugge 2015 ESA Mulier 2015 www.ESPCOP.org 36
- 38. 7th ESPCOP Meeting December 17th 2016 View publication statsView publication stats
